Thyroid hormone (TH) is an important signalling molecule that regulates the behaviour of various cells, tissues and organs throughout the body. Under normal conditions, it is produced by the thyroid gland, and its concentration throughout the body is maintained by the hypothalamic-pituitary-thyroid axis.
Excess TH may manifest in accelerated systemic metabolism and several disorders known as thyrotoxicosis.1,2 Its most severe form is called thyroid storm (TS).2,3 Although the incidence of thyrotoxicosis accompanied with TS is relatively rare, it is associated with a higher mortality rate (1.2–3.6%) than thyrotoxicosis without TS (0.1–0.4%).4 The most common aetiology of thyrotoxicosis is Graves’ disease (GD), followed by nodular thyroid disease and thyroiditis.5
Life-threatening complications of thyrotoxicosis can have cardiac manifestations. It can manifest as severe as an arrhythmia leading to heart failure (HF), or have less severe manifestations such as sinus tachycardia and AF.6 These conditions are facilitated by alterations in cardiovascular function, including increases in heart rate (HR), systolic blood pressure (SBP), left ventricular (LV) contractility, blood volume, myocardial oxygen consumption and cardiac output (CO), followed by decreases in systemic vascular resistance (SVR) and diastolic blood pressure (DBP).6,7 Pulmonary artery pressure (PAP) is also elevated in hyperthyroidism, resulting in pulmonary hypertension (PH). These altered cardiovascular states may lead to the exacerbation of existing valve conditions, namely tricuspid regurgitation (TR) and mitral regurgitation (MR), or trigger novel valve disorders.7
Valvulopathy in thyrotoxicosis, especially TR and MR, is a consequence of the change in cardiac pressure and volume overload; however, the precise mechanism and reversibility of thyrotoxic valvulopathy remain unclear.8 As yet, there are no clear data on thyrotoxic TR and MR, including on the clinical characteristics and survival.
Regarding the statements above on the rarity of the condition, we aim to summarise the findings from case reports through a systematic review. The primary outcome is the progression or improvement of TR and/or MR through echocardiographic evaluation after thyrotoxicosis treatment, while the secondary outcomes are progression or improvement of HF and PH.
Methods
This systematic review was conducted in accordance with PRISMA 2020.9 The protocol of this study was registered in the International Prospective Register of Systematic Reviews (PROSPERO) database (CRD42023462255).
Search Strategy
We conducted a systematic search for relevant studies in five electronic medical databases – PubMed, Scopus, Web of Science, ProQuest and Taylor & Francis (www.tandfonline.com/search/advanced) – from inception to 17 June 2023. The keywords in each database were based on Medical Subject Headings terms and combined with several additional terms (Supplementary Table 1). Boolean operators were used to broaden and narrow the search results. The search was restricted to human participants with no language and publication date restrictions.
Eligibility Criteria
Studies were included if they met the following inclusion criteria: study design of case reports or case series; and participants were adult patients aged ≥18 years diagnosed with thyrotoxicosis, impending TS or TS accompanied by TR and/or MR. The measured outcomes were the number of TR and/or MR events, any echocardiographic findings (i.e. valvular regurgitation severity, LV ejection fraction (LVEF), evidence of PH and its severity or evidence of heart chamber enlargement or dilation), clinical presentations, thyroid function test results and other thyroid-related parameters, and/or pharmacological and non-pharmacological treatment approaches.
Several exclusion criteria were applied as follows: duplicate studies; irrelevant titles and/or abstracts; irretrievable full texts; inappropriate study design (e.g. review articles, observational studies, systematic reviews, meta-analyses, letters to editors and conference abstracts); and an inappropriate population, including pregnant women, patients with valve-related comorbidities or other comorbidities that might interfere with heart function (rheumatic heart disease, pericarditis, myocarditis, endocarditis, uncorrected or major congenital heart disease, acute coronary syndrome, pulmonary embolism, previous cirrhosis or heavy alcoholic drinking), and people with dominant hyperthyroidism-unrelated signs and symptoms at the first presentation or unexpected findings of hyperthyroidism.
Data Extraction and Quality Assessment
Six of the authors independently screened all relevant studies, and any disagreements were resolved by all authors reaching a consensus.
Several data were extracted from the included studies, including: sex, age, study location (country and region), follow-up duration, underlying disease and its status on admission (i.e. thyrotoxicosis, impending TS and TS) based on the Burch-Wartofsky Point Scale (BWPS) or the authors’ statements where BWPS was not reported; vital signs (i.e. SBP, DBP, HR and palpatory rhythm), clinical presentations and assessments (i.e. history-taking, physical examination, clinical evidence of HF and clinical outcomes), thyroid function test results and other thyroid-related parameters (i.e. thyroid-stimulating hormone or thyrotropin, free thyroxine, free triiodothyronine, thyroid-stimulating hormone receptor antibody, thyroid-stimulating antibody or immunoglobulin or thyroid-stimulating immunoglobulin, thyroglobulin antibody, thyroid peroxidase antibody or anti-thyroid microsomal antibody); echocardiographic methods and findings (i.e. TR and MR severity, LVEF, evidence of PH and its severity, evidence of heart chamber enlargement or dilation and severity); and pharmacological and non-pharmacological treatment approaches.
The quality assessment of the included studies was performed using the latest scoring system for case reports and case series proposed by Murad et al., as modified by Smith et al. and Wang et al.10–12 The quality of the studies was considered good (low risk of bias) when five criteria were met, moderate when four criteria were fulfilled and poor (high risk of bias) when three criteria or fewer were met.12
We performed the data extraction and the quality assessment of the included studies through collaborative group discussion, and final decisions were reached with all authors’ agreement.
The reported clinical outcomes were separated into two groups: condition on discharge; and final thyroid status at the last follow-up. Then we applied a classification for each group. The condition upon discharge was categorised as follows: improved/recovered; improved followed by readmission; improved with any persistent conditions; died; and unclear. The final follow-up of thyroid status was categorised as follows: euthyroid, on medication; euthyroid, off medication; euthyroid, unclear medication status; uncontrolled, poor compliance; lost to follow-up; and unclear.
The echocardiographic severity of TR and MR was classified as the following: trace/trivial; mild; mild to moderate; moderate; moderate to severe; severe; and very severe. This classification was drawn up based on the direct statements in each study without any reported parameters.
Echocardiographic evidence of HF was classified based on the reported LVEF: HF with reduced EF (HFrEF) as LVEF ≤40%; HF with mildly reduced EF (HFmrEF) as LVEF 41–49%; and HF with preserved EF (HFpEF) as LVEF ≥50%.13
PH severity was classified into the following consecutive grades: mild; moderate; and severe. PH severity classification was based on any of the following reasons: direct statements with no reported parameters; systolic PAP if reported, then classified into mild (35–50 mmHg), moderate (50–70 mmHg) and severe (>70 mmHg); or right ventricular (RV) systolic pressure if reported, then classified into mild (35–45 mmHg), moderate (46–60 mmHg) and severe (>60 mmHg).14,15
Statistical Analysis
The descriptive statistics were analysed using IBM SPSS (version 25.0; IBM). The overall results were presented in three main groups: TR only; MR only; and both. The categorical data were the number of events (n) and the percentages (%), while the continuous data were presented according to the Kolmogorov-Smirnov normality test.
The data were presented as means ± standard deviation (SD) if p≥0.05; otherwise, they were presented as medians (interquartile range). We also created a follow-up trend analysis graph on the improvement of TR and/or MR severity, LVEF as well as PH severity using a spreadsheet (version 16; Excel, Microsoft).
Results
Literature Search and Included Studies
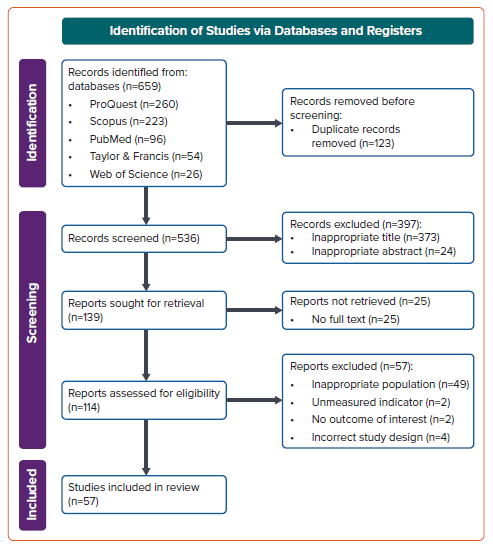
Our initial search yielded 659 studies, of which 123 were removed as they were duplicates. We obtained 139 studies with appropriate titles and abstracts, and removed 25 of these as the full text was not available. After applying the inclusion and exclusion criteria to the remaining 114 studies, 57 eligible case reports and case series with a total of 62 eligible patients were included in this study (Figure 1).
The summary of the qualitative synthesis is provided in Supplementary Data 1. The study quality assessment showed that 33 studies had a low risk of bias, 24 studies had a moderate risk of bias, and no studies had high risk of bias (Supplementary Table 2).
Baseline Characteristics
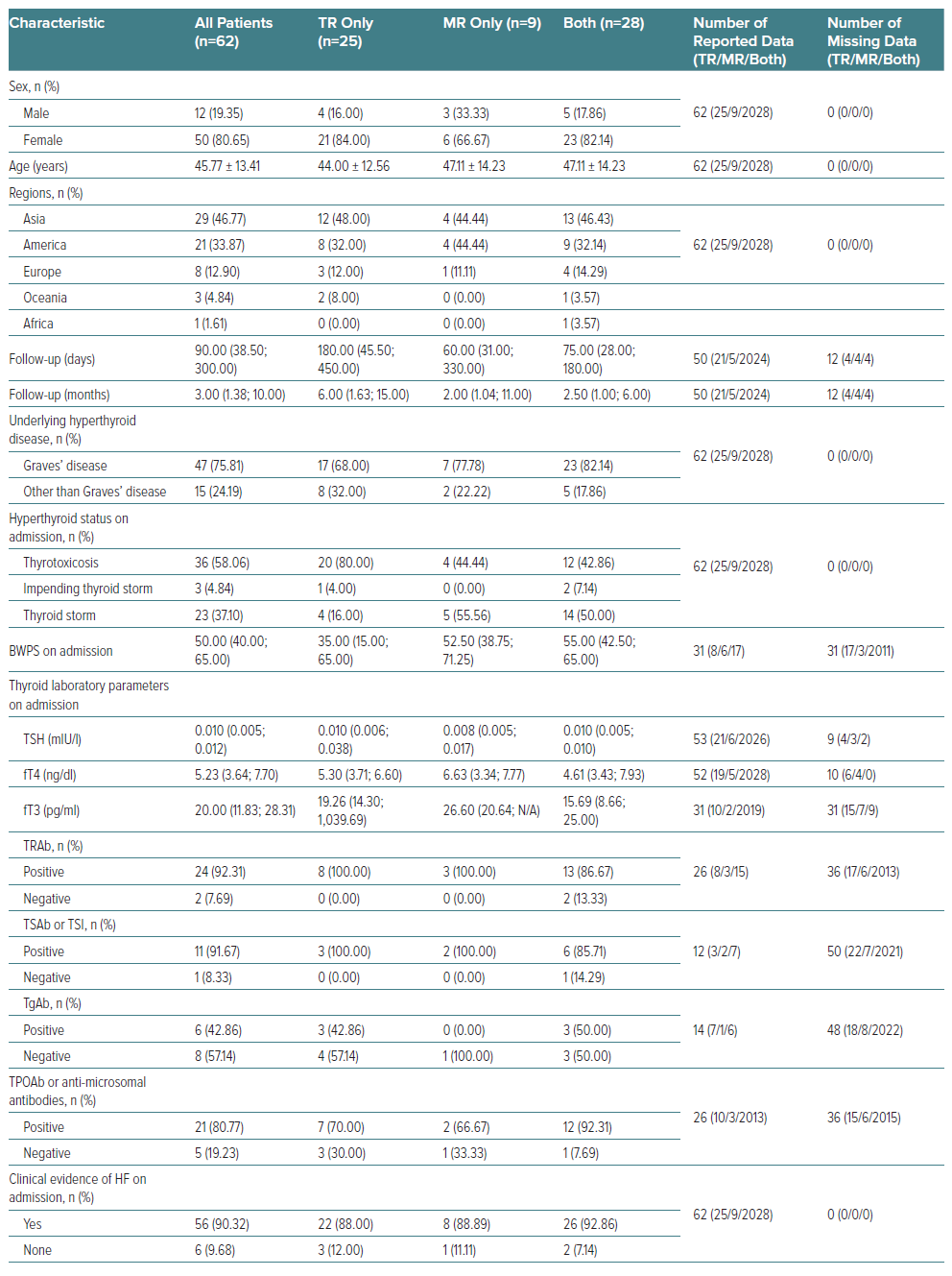
The overall basic characteristics of the eligible patients on admission are provided in Table 1. The eligible patients in our study were mainly women (n=50; 80.65%) and the mean age was 45.77 years. Participants were mostly from Asia (n=29; 46.77%) and America (n=21; 33.87%); this geographical area also applied to each group of TR only, MR only and both.
The longest follow-up period was found in TR-only group, with a median of 6 months or 180 days. GD was the most common underlying disease in all patients, affecting approximately three-quarters of them. All patients were diagnosed with thyrotoxicosis, with 23 of them (37.10%) having TS. Approximately half of the patients with MR, whether MR only or with both valves affected, had TS. The median BWPS values were 35.00 (15.00–65.00) for TR only, 52.50 (38.75–71.25) for MR only and 55.00 (42.50–65.00) for both valves affected. The overall thyroid-stimulating hormone, free thyroxine and free triiodothyronine values showed that all patients were experiencing primary hyperthyroidism. On the final follow-up of thyroid status, most patients (n=40; 64.52%) were euthyroid regardless of their treatment status; this finding was similar in each group. Three patients had hypothyroidism: one had undergone total thyroidectomy and two had been given radioactive iodine ablation.
Two clinical factors were also included as basic characteristics of the patients: clinical evidence of HF on admission; and clinical condition after the first discharge. Of the 56 patients who had clinical evidence of HF on admission, almost half of them (n=27; 48.21%) had isolated right HF (RHF); they accounted for 17 out of 22 TR-only patients (77.27%), no patients in the MR-only group and 10 of the 26 patients with both valves affected (38.46%).
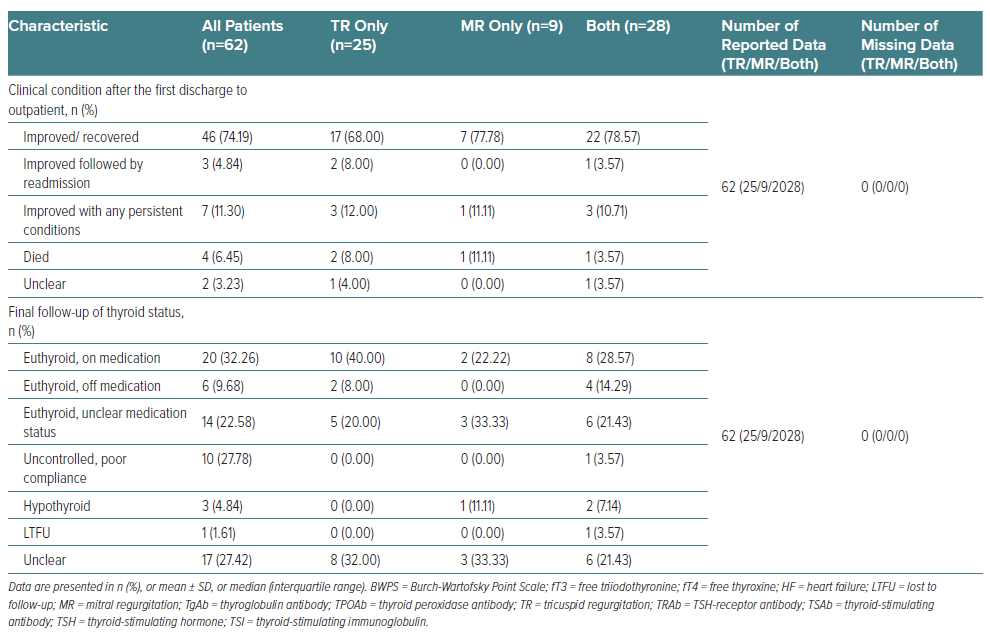
Patients had mostly improved or recovered at their first discharge. However, three (4.84%) were readmitted once after their first discharge: one patient with both valves affected then died in hospital; and the two others (who were in the TR-only group) recovered on the second discharge.16–18 Seven patients (11.30%) had clinical improvement on their first discharge but still had persistent conditions that could not be fully healed during their inpatient period, including lower limb oedema, cardiomegaly/cardiomyopathy, AF, persistent hypocalcaemia and hypoalbuminaemia, cirrhosis, ascites and PH.19–25
The clinical presentation of the patients on admission based on history taking and physical examination is shown in Supplementary Table 3. The most common hyperthyroidism-related complaints were general dyspnoea (n=34; 54.84%) followed by palpitations (n=33; 53.23%). These findings were also found in the TR-only and MR-only groups. The most common hyperthyroidism-related complaints in patients with both valves affected were dyspnoea on exertion (n=16; 57.14%) and general dyspnoea (n=15; 53.57%). Chest pain, cough and abdominal pain (each n=6; 9.68%) were the most common complaints unrelated to hyperthyroidism.
On physical examination, the MR-only patients had the highest median HR of 138 BPM of all groups. More than half of the patients (n=38; 65.52%) had irregular heartbeats. Head and neck examination showed that goitre (n=39; 62.90%) and elevated jugular venous pressure (n=32; 51.61%) were the two most frequent findings. Most patients with TR had an elevated jugular venous pressure, either TR only (n=17; 68.00%) or both valves affected (n=14; 50.00%). Anaemic conjunctivae (n=6; 21.43%) and icteric sclerae (n=8; 28.57%) were found more in the groups with both valves affected than in single-valve groups. In the thorax examination, systolic murmur was the most frequent finding (n=37; 59.68%).
There were signs of lung congestion, mostly found in patients with MR, whether MR only (n=2; 22.22%) or with both valves affected (n=9; 32.14%). In the abdominal examination, signs of ascites and congestive hepatopathy/cardiac cirrhosis were both most commonly found in patients with TR, whether TR only (each n=6; 24.00%) or both valves affected (each n=6; 21.43%). The most frequent findings in the extremities examination were bilateral lower oedema (n=38; 61.29%) and tremors (n=12; 19.35%).
We categorised the treatment approaches into pharmacological and non-pharmacological. We further classified pharmacological treatments based on the drug class (Supplementary Table 4). On the first presentation of 62 patients, 11 (17.74%) received propylthiouracil (PTU), 40 (64.52%) received methimazole (MMI) or its derivatives, one (1.61%) received both, one (1.61%) received Lugol’s iodine without a thionamide, two (3.23%) received unspecified antithyroid drugs (ATDs), and seven patients (11.29%) did not receive any ATDs. For the HR and oedema controls, the patients were given β-blockers and diuretics, with propranolol (n=30; 48.39%) and furosemide (n=24; 38.71%) the most commonly used, respectively.
To prevent further deterioration, several patients were given angiotensin-converting enzyme inhibitors (ACEis) (n=9; 14.52%) or angiotensin receptor blockers (ARBs) (n=2; 3.23%). Patients also received corticosteroids as adjunctive therapy. The most frequently used were the short-acting corticosteroids (n=11; 17.74%), followed by intermediate-acting (n=8; 12.90%) then long-acting (n=2; 3.23%). Other pharmacological treatments were used supportively to maintain their clinical conditions, including cholestyramine (n=5; 8.06%).
Non-pharmacological treatments were varied; eight patients (12.90%) had radioactive iodine ablation, four patients (6.45%) received therapeutic plasma exchange or plasmapheresis, five patients (8.06%) had valvular repair or replacement surgery, and five patients (8.06%) underwent thyroidectomy.
Echocardiography
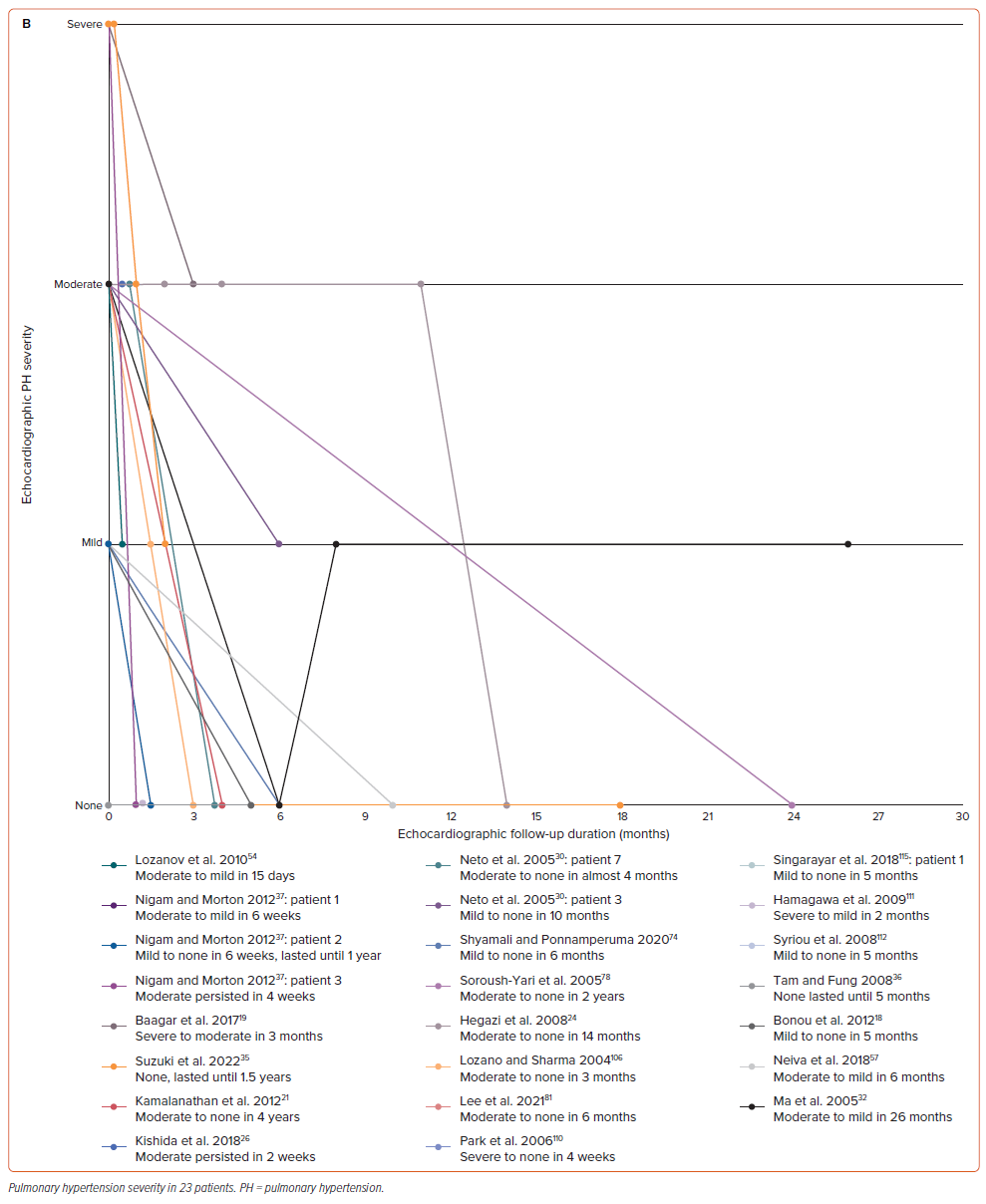
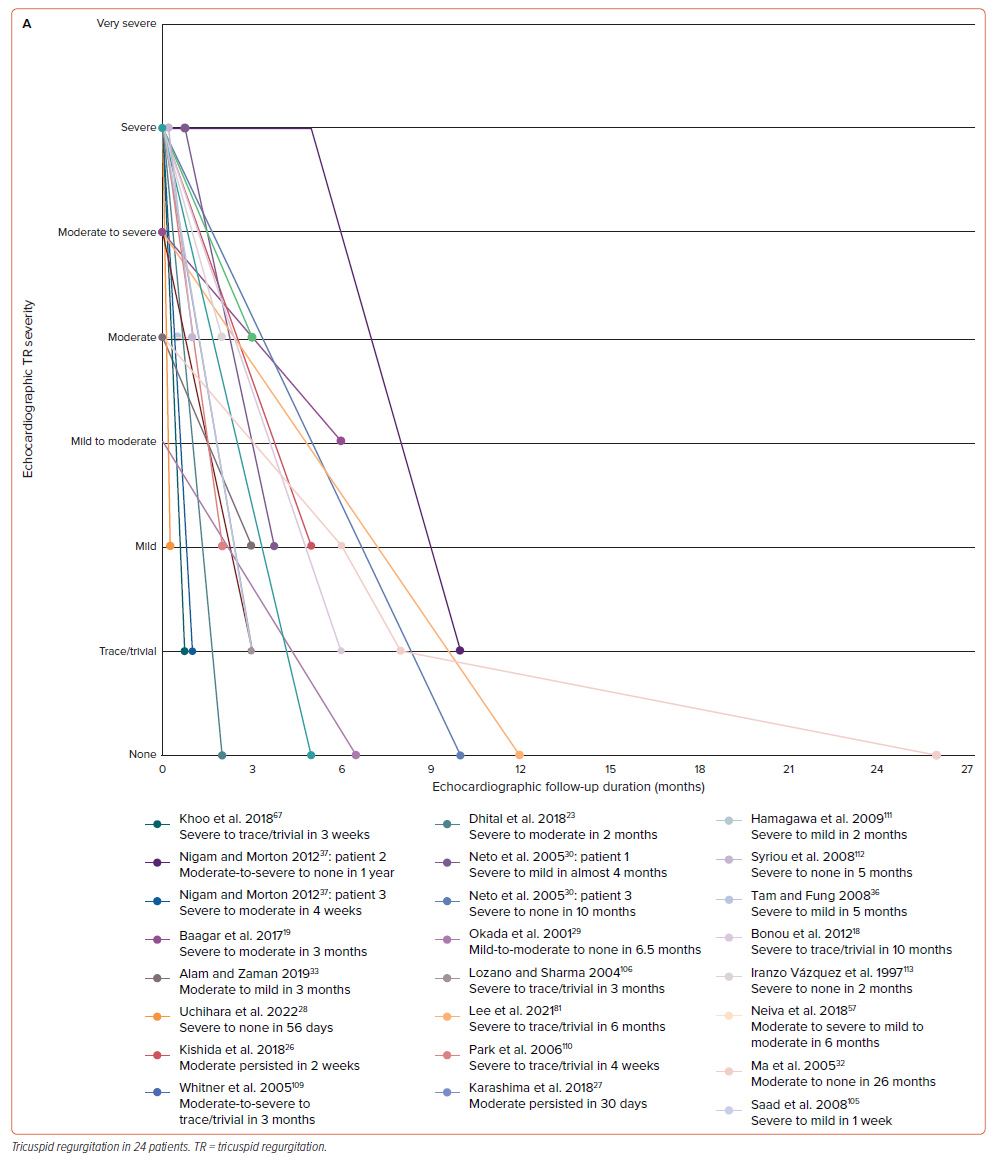
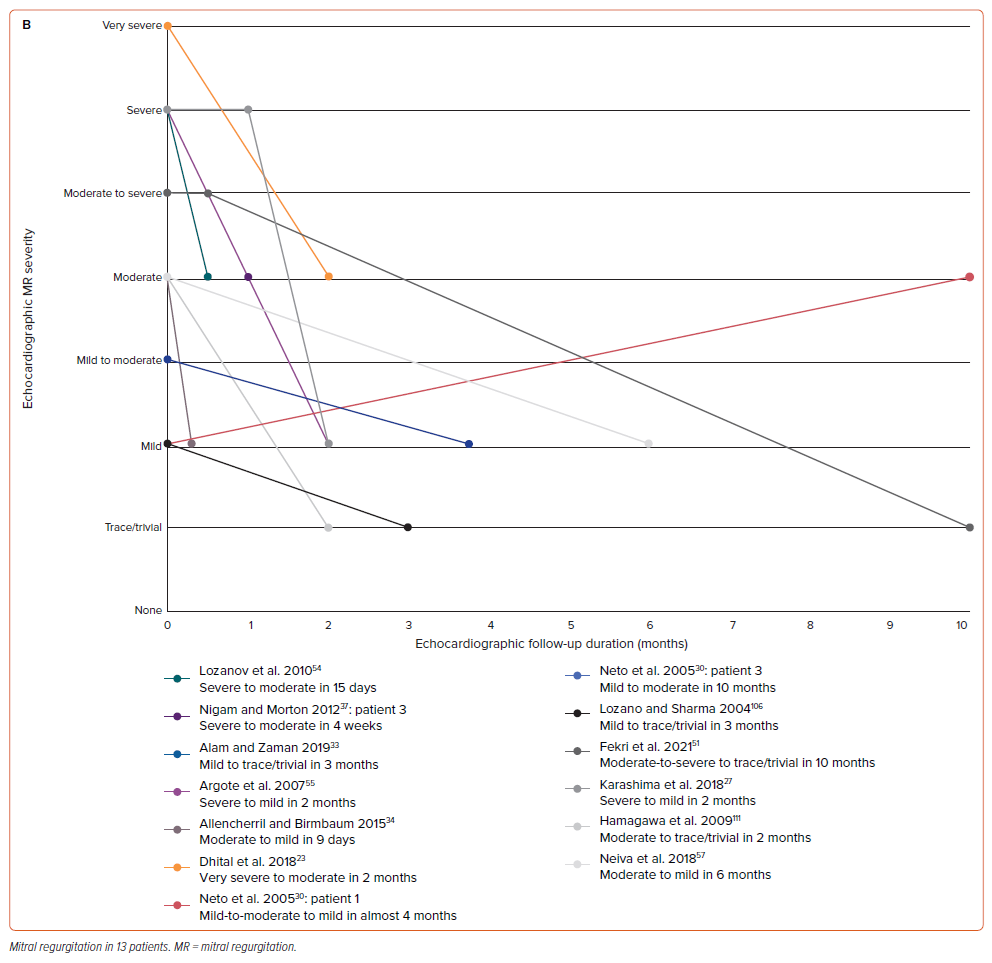
A summary of echocardiographic results on admission is presented in Supplementary Table 5. Most patients were examined using transthoracic echocardiography (TTE) (n=53; 85.48%). In comparison to the other severity, we discovered that severe and moderate TR were the most common types (n=26; 50.98% and n=13; 25.49%, respectively). A similar finding was also observed in the MR group regarding its severity (each n=10; 27.78%). However, a very severe degree of valvular regurgitation was found in two patients with MR when concomitantly occurring with TR. We further observed the progression of TR and MR in several patients by showing follow-up graphs (Figure 2). In patients with TR (Figure 2A), the improvement could be seen in the first 6 months of follow-up, which was found in 20 out of 24 patients (83.33%). One patient still had persistent moderate TR after two weeks and another after 30 days of follow-up; however, the follow-up data after those periods were not provided for both patients.26,27 The complete TR disappearance was found in five patients.28–32 In patients with MR (Figure 2B), improvement could be seen in the first three months of follow-up in nine out of 13 patients (69.23%). However, there were some differences in progression between MR and TR. One patient progressed from mild to moderate and none progressed to the complete MR disappearance during the follow-up period.30
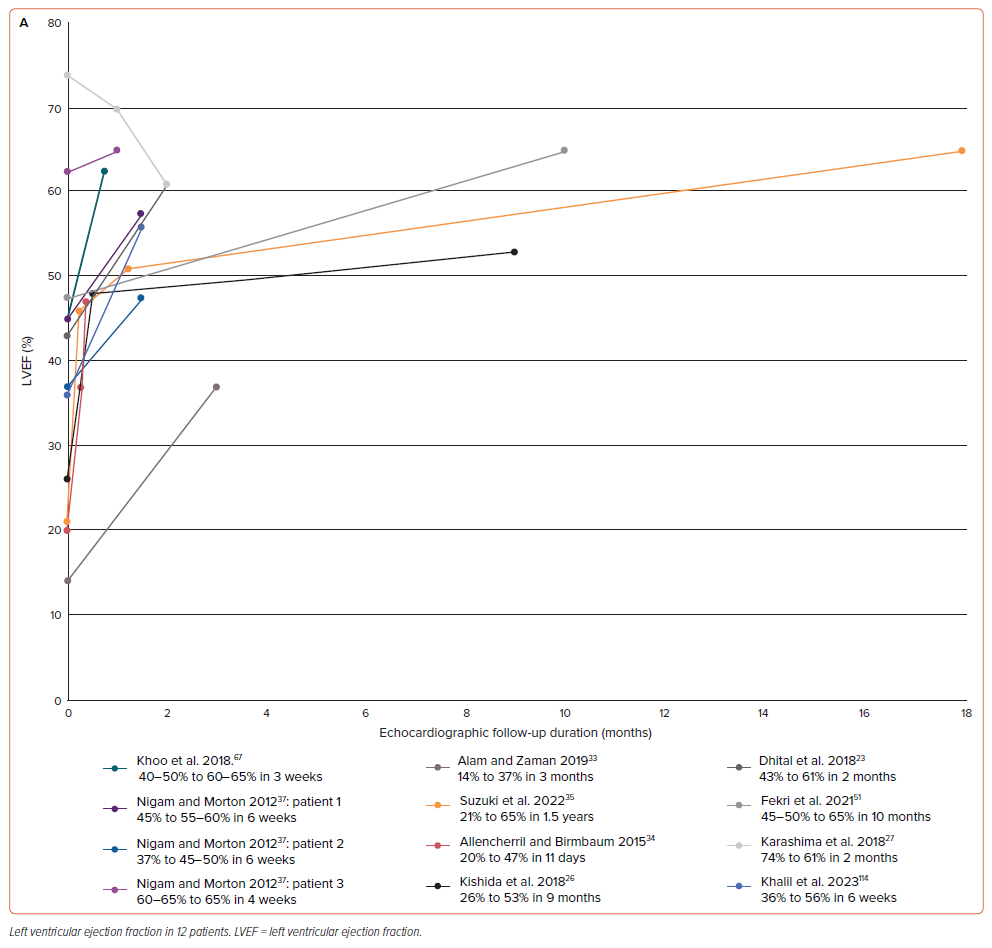
Furthermore, HFpEF was the most common type of HF (n=20; 45.45%), followed by HFrEF (n=12; 27.27%). In a further observation, the most frequent type of HF in TR only was HFpEF (n=12; 70.59%), while that in MR only was HFrEF (n=5; 71.43%). We also observed cardiac function progression through the LVEF follow-up graph (Figure 3A). LVEF improvement appeared in the first two months of follow-up in eight out of 12 patients (66.67%); two of those eight patients still had an LVEF <50% during the first two months of follow-up.33,34 However, while 11 of 12 patients saw LVEF increase during the follow-up period, one experienced a decrease in LVEF.27
Regarding the PH severity, moderate PH was the most common overall, especially in the TR group (n=11; 45.83% in TR only and n=10; 43.48% in both valves affected). This finding was similar in severe PH. However, there were three patients whose PH severity was not described. Similar to valvular regurgitation and HF, we created a follow-up graph for PH progression (Figure 3B). Two patients were without PH from presentation to the end of the follow-up period, leaving 21 patients with PH.35,36 Improvement in PH was mostly seen in the first 6 months of follow-up in 16 of 21 patients (76.19%). Two patients had persistent, moderate PH during the first month of follow-up; however, neither had any further echocardiographic PH follow-up results.26,37 Throughout the whole follow-up period, the PH disappeared in 13 out of 21 patients (61.90%). One patient experienced an improvement from moderate to complete PH disappearance, then progressed to mild PH.32
Our echocardiographic summary also included evidence of atrial and ventricular enlargement or dilation. Right atrial enlargement or dilation was found mostly in the TR-only group (n=9; 81.82%). In patients with both valves affected, both atria were commonly enlarged (n=14; 66.67%). Moreover, RV enlargement or dilation was commonly found in the groups with TR, in both the TR-only group (n=15; 78.95%) and in those with both valves affected (n=12; 66.67%).
Discussion
In this study, the 62 thyrotoxic patients in the 57 studies were predominantly women. This is consistent with previous studies that have shown thyrotoxicosis is more common in women than men.4,38,39 This may be owing to the autoimmune nature of thyroid diseases, secondary to the high oestrogen and progesterone levels.40 TR and MR are increasing in females and occur more commonly in women than men.41–44 Although our findings are consistent with previous research, we need to acknowledge that our study participants are predominantly women, which influences the number of thyrotoxic TR and MR cases. Nonetheless, both sexes have their own susceptibilities to TR and MR because of differences in basic anatomical structures.44
Based on the mean age, the included patients are in their mid-to-late 40s. Age is an important factor in the development of thyrotoxic TR and MR, since the incidence and severity of valvular regurgitation increase with age owing to mechanical wear and tear.41,43 Furthermore, most patients in each valvular group have clinical evidence of HF, such as peripheral oedema, exertional dyspnoea or tachycardia.45 HFpEF and HFrEF were most commonly found in the TR-only and MR-only groups, respectively, which is consistent with previous research.46 In addition, one study states that bivalvular regurgitation is prevalent among HFrEF in the general population.47 This slightly different finding indicates that thyrotoxicosis, as an underlying disease, may affect the prevalence of HF in a population. Regarding PH, moderate and severe PH are both commonly found in patients with TR. It indicates that thyrotoxic TR can be caused by hyperthyroidism-related PH, although the exact mechanisms are still not fully understood.48,49 Therefore, we will comprehensively review the possible mechanisms of thyrotoxic TR and/or MR related to hyperthyroidism-related HF and PH in the next section.
Thyrotoxic Tricuspid and Mitral Valve Regurgitation Pathophysiology
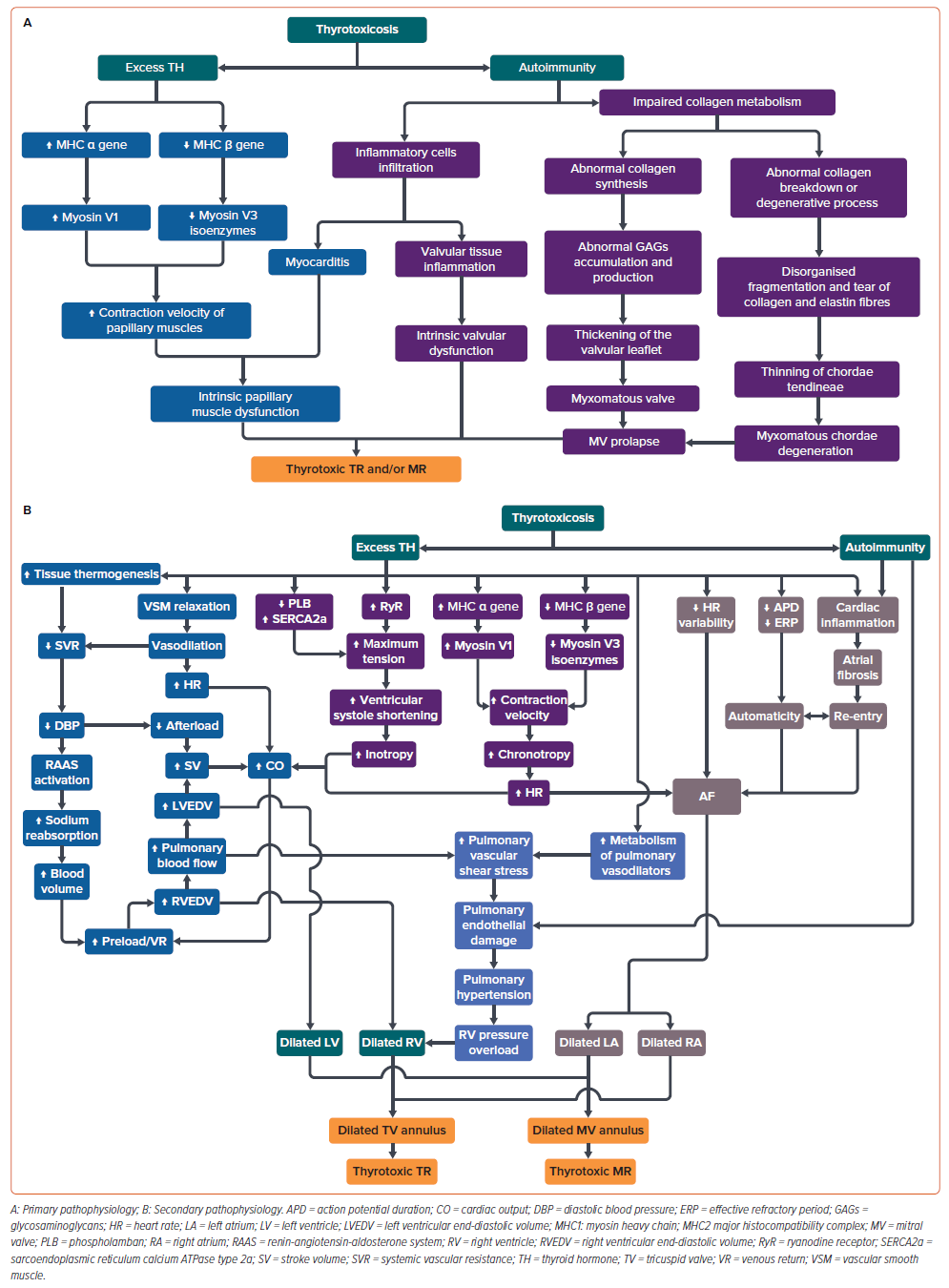
Thyrotoxic TR and/or MR are assumed to be caused by two groups of mechanisms: primary valvular dysfunction and its apparatus (i.e. chordae tendineae and papillary muscles); and secondary valvular dysfunction due to the haemodynamic effects of circulating TH.50,51 These mechanisms, especially degeneration of the valve leaflet, may affect the chordae tendineae, which cause the structure to thicken, elongate and rupture, which is then followed by permanent damage to the valve leaflet.52
The mechanisms involved in primary valvular dysfunction, whether TR or MR, arise mainly from the direct effect of excess TH and autoimmunity in thyroid diseases (Figure 4A).51,53,54
Excess TH may induce intrinsic papillary muscle dysfunction or thyrotoxic myopathy, which is mediated through the direct TH effect on cardiac muscles.8,53,55 TH upregulates the myosin heavy chai α gene transcription to increase myosin V1 with higher ATPase activity and downregulates the myosin heavy chain β gene transcription to decrease myosin V3 isoenzymes. This process leads to a faster movement of globular myosin head along the thin filament and increases the contraction velocity of the papillary muscles, which may cause them to fatigue easily or experience a transient dysfunction.8,56,57
Furthermore, the autoimmunity in thyroid disease may trigger two processes. Autoimmunity may cause inflammation in either the valve itself or its papillary muscles due to lymphocytic infiltration.51,58,59 An impairment in collagen metabolism in the valvular structure with its chordae tendineae is observed in autoimmune thyroid disease, known as myxomatous changes, which is the main cause of MV prolapse (MVP).23,27,50
Abnormal collagen synthesis – abnormal accumulation and production of glycosaminoglycans – is more commonly found in the valvular structure, leading to the valvular leaflets thickening. This process is similar to the pathophysiology of exophthalmos and pretibial myxoedema.27,51
The pathophysiology involved in the chordae tendineae is more of a degenerative process or collagen breakdown, which leads to the disorganised fragmentation and tearing of collagen and elastin fibres histology shows.27,51,60 This leads to easily stretched and ruptured chordae tendineae at a lower shear stress than normal tissue.50
Even though these pathophysiological processes in thyrotoxicosis are more investigated in MV than TV, similar processes are thought to be involved in TV.50
Interestingly, the mechanisms involved in secondary thyrotoxic TR and/or MR are also related to the excess T3 and autoimmunity (Figure 4B). However, autoimmunity does not play a significant role as in the primary dysfunction. The first pathway – the non-genomic pathway – is through the effects of T3 in increasing the bodily tissue thermogenesis and inducing the relaxation of vascular smooth muscles, which both cause a decrease in SVR and an increase in HR due to vasodilation.61–65 Then, the low SVR induces DBP reduction, while the tachycardia increases the CO.61,66
The DBP reduction causes a split in the pathway; it directly lowers the cardiac afterload and activates the renin-angiotensin-aldosterone system (RAAS), which increases the preload/venous return due to an increase in blood volume, followed by increases in biventricular end-diastolic volumes.61,63,67 Both latter processes (decrease in cardiac afterload and activated RAAS) then raise the CO.23,61,63,64,66,67 If this continues, a vicious cycle may be established and cause RV and LV dilation. It may give rise to the dilation of atrioventricular valve annulus and the thyrotoxic TR and/or MR appearance due to the incomplete leaflet coaptation.23,50,51
This pathway is supported by other non-genomic effects of T3: it reduces the HR variability, action potential duration, and effective refractory period of the cardiomyocytes; and induces cardiac inflammation by promoting myocardial nitric oxide synthase activity and increasing tumour necrosis factor α, interleukin-1, interleukin-6 and interferon γ.68,69
Cardiac inflammation can be exacerbated by the autoimmunity of thyroid disease and may result in fibrosis of atrial tissue.58,68 This may cause AF through a back-and-forth phenomenon of automaticity and electrical re-entry.68 If the AF is left untreated, this may progress to both right and left atrial enlargement, which have been proven to cause annular dilation of the atrioventricular valve and may cause thyrotoxic TR and/or MR.70–72
The second pathway, or the genomic pathway, is through the effects of T3 (Figure 4B). It induces the upregulation of the myosin heavy chain α gene and the downregulation of β gene expression, which increases the cardiomyocytes’ contraction velocity.8 This affects cardiac chronotropy, which influences the non-genomic pathway by increasing CO and the likelihood of AF.62,64
T3 also upregulates the expression of ryanodine receptors and sarcoendoplasmic reticulum calcium ATPase type 2a, and downregulates the phospholamban expression, a sarcoendoplasmic reticulum calcium ATPase inhibitor. This process affects cardiac inotropy, increasing the maximum tension of cardiac myofibrils and their shortening in the ventricular systole phase.62,66 CO then also increases in response to the powerful cardiac contraction.
All genomic pathways will also affect the non-genomic pathway by causing dilation in the atrioventricular valve annuli through dilation of the atrial and ventricular chambers.
The other secondary pathways are specifically for thyrotoxic TR only (Figure 4B). The association between hyperthyroidism and PH has been established. The underlying pathophysiology of PH in hyperthyroidism is still unclear, although several mechanisms have been hypothesised.73,74
First is autoimmune-mediated endothelial damage. In autoimmune hyperthyroidism (GD), autoantibodies have been suggested as the primary cause of PH. Autoantibodies may injure pulmonary vascular endothelial cells, leading to elevations of pulmonary vascular resistance and, subsequently, the development of PH. This autoimmune-related mechanism is supported by the observation of neonatal thyrotoxicosis in infants born to mothers with GD, which in most cases are due to transplacental passage of the maternal thyroid-stimulating immunoglobulin. Cases of neonatal thyrotoxicosis complicated with PH have been reported, and all showed a complete reversal of PH after the thyrotoxicosis was treated.75,76
Another hypothesis is mechanical/high CO-mediated endothelial dysfunction. The effects of excess TH on the cardiovascular system have been well described.62 TH directly enhances cardiac contractility, reduces SVR via vasodilation of arterioles and raises circulating blood volume via RAAS activation and increased erythropoietin secretion, which increase the RV preload and CO. The resultant hyperdynamic circulation and high CO state cause a substantial flow shear stress upon the pulmonary vascular beds. This mechanical stimulus increases intracytoplasmic calcium concentration in pulmonary arterial smooth muscle cells that induces vasoconstriction, consequently increasing PAP.77
Altered metabolism of pulmonary vasoactive substances could also be a mechanism. In hyperthyroidism, metabolism of intrinsic and extrinsic pulmonary vasodilators, such as NO, prostacyclin, and acetylcholine, is accelerated alongside inhibited metabolism of pulmonary vasoconstrictors, such as endothelin-1, serotonin and thromboxane.23 These lead to further increases in pulmonary vascular resistance.
Finally, sympathovagal imbalance is suggested to underlie the pathophysiology of PH. Greater sympathetic stimulation and reduced cholinergic response secondary to hyperthyroidism, respectively, causing increased sympathetic tone and decreased vagal tone, are suggested to induce pulmonary vasoconstriction.30,78,79 This PH then leads to the development of functional TR; their association is widely known. Functional TR arises due to RV pressure overload, followed by structural alterations in the right heart myocardial geometry and RV dysfunction, causing tricuspid annulus dilation and leaflet tethering.80 However, TR may also be directly related to thyrotoxicosis independently from the presence of PH.18
Time- and Treatment-Related Reversibility of Thyrotoxic Tricuspid Regurgitation and/or Mitral Regurgitation
Following medical treatment of thyrotoxicosis-related valvular regurgitation (involving ATDs, β-blockers, ACE-I/ARB and diuretics), several studies showed that once a euthyroid state is achieved after thyrotoxicosis, cardiac function returns to normal within 6 months of therapy.81–85 This is parallel to our findings, which found an improvement in overall cardiac function – decreased regurgitation severity, increased LVEF, reduced systolic PAP – within the first 2–6 months of therapy. Complete cardiac function recovery within 6 months of thyrotoxicosis is dependent on the pre-treatment disease duration, β-blocker administration and whether a euthyroid state is achieved.82,86 As shown in the results, several patients experience persistent moderate TR, MR and/or PH despite aggressive treatments.26,27,37 This can be explained by the fact that the parameters for TR, MR and/or PH are followed up only in their first month of therapy, while in general, those parameters usually improve in the first 3–6 months of therapy. This is supported by previous statements regarding the improvement of cardiac function within 6 months of therapy.81–83 Therefore, the timing of echocardiographic follow-up is important to determine the cardiac function improvement in thyrotoxicosis.
ATDs, such as MMI and its derivatives, have been shown to prevent cardiac hypertrophy and mortality in animal research.87 A recent study also suggests the cardioprotective effect of ATDs in that MMI may include mitigating arrhythmia in thyrotoxic patients.88 PTU may also provide protection against oxidative stress due to increased TH levels.89,90 This supports our study that the two most commonly used ATDs are MMI or its derivatives and PTU as the pivotal treatments. Oxidative stress can affect the cardiovascular system, including the cardiac valves.91 It may exacerbate and cause damage to the myxomatous valve leaflet degeneration further to that caused by thyrotoxicosis.51 Hagler et al. reported that oxidative stress contributes to the myxomatous MV degeneration.92 Therefore, ATDs may play a part in preventing and treating valvular heart diseases and valve-related complications by helping the body to maintain normal TH levels.
TH also upregulates β 1 adrenergic receptors on the heart, affecting various cardiac haemodynamic components such as HR, BP, cardiac muscle contractility and CO. During thyrotoxicosis, cardiac function is heightened due to increased TH levels.93 β-blocker administration may diminish the excessive effects of TH on cardiac β 1 adrenergic receptors.94 β-blocker administration in patients with MR is proven to increase their future survival and reduce the regurgitant volume and LV workload.95,96 Unfortunately, those effects are still unknown in TR as studies are limited. Aside from those effects, β-blockers, mainly propranolol, are still effective in blocking the peripheral conversion of T4 to T3, which promotes the normalisation of TH levels and may ameliorate thyrotoxic valvulopathy.94
Previous research has shown that the administration of ACEis/ARBs in patients with MR may improve the regurgitant fraction and volume, as well as LV size.84 Another study has also shown that ACEi use reduces TR-related HF symptoms.85 TH may affect the local RAAS on the human heart, which expresses angiotensin II type 1/2 receptors. During the elevated TH levels, renin production on local RAAS increases, followed by the production of angiotensin II and its receptors. Angiotensin II promotes cardiac hypertrophy and fibrosis through those receptors.97 These mechanisms may be mitigated by ACEis/ARBs, which help cardiac function to cover and prevent cardiac tissue fibrosis and hypertrophy. Furthermore, ACEis/ARBs can be a conservative treatment for moderate-to-severe MR to lower the risk of MR-related all-cause death and HF-related hospitalisation and readmission.98
There are several distinctive supportive treatments given to improve the thyrotoxicosis and the valvular dysfunction.
First, the use of corticosteroids in thyrotoxicosis or TS is still controversial.99 In two cases, dexamethasone was used as the long-acting corticosteroid to control/reduce the intensity of thyrotoxicosis and to prevent further complications.53,73 Furthermore, the intermediate-acting corticosteroids (prednisone, prednisolone and methylprednisolone) were administered in eight cases for several reasons: to treat severe hepatic dysfunction; to prevent possible contrast-related allergic reactions; to pre-medicate the patients before surgery and cardiac catheterisation; and to reduce free thyroxine levels before radioactive iodine treatment.29,37,65,100–102 However, no specific reasons were observed in the 11 cases where hydrocortisone was the short-acting steroid.8,16,25,35,53,67,100,103–105 Due to the differences of treatment goals using corticosteroids, further studies are warranted to establish the efficacy of corticosteroids in thyrotoxicosis or TS. Its use is still dependent on the individual patient basis and the doctor’s clinical judgement.99
Second, cholestyramine was given in five cases as an alternative to treat patients with multiple complications while trying not to aggravate pancytopenia and as an adjuvant therapy due to its benefit as a conventional therapy.19,25,53,57,102
Third, four cases were treated using therapeutic plasma exchange based on several reasons: to remove excess TH, toxins from liver dysfunction TSAb, and catecholamines, along with reducing TH bound to thyroid-binding globulin, transthyretin and plasma albumin; to ameliorate severe hepatic failure; to avoid haemodialysis in refractory TS; and to improve overall clinical manifestation.35,65,102,103
Fourth, the valvular repair or replacement was performed in five cases due to MV and TV leaflet prolapse, annular dilatation and chordae tendineae rupture, to accelerate the overall clinical improvement after refractory hypotension and overt HF.17,27,51,53,60
Lastly, thyroidectomy was performed in five cases as a definitive therapy to accelerate the improvement of refractory hyperthyroidism and thyrotoxic valvulopathy, especially after the worsening clinical conditions despite adequate treatments.27,29,37,106,107
Limitations
There are several limitations in our study. First, this systematic review includes case reports as reported cases are rare. Case reports are inherently anecdotal, provide a specific non-random sample and may not allow for claims of causality.11,108
Second, the case distribution is dominated by Asia and America; therefore, it is possible that similar cases in other continents are underreported.
Third, the grading of TR and MR severity is based on the authors’ eyeball findings from the echocardiography, which is an operator-dependent examination.
Fourth, data in some case reports are incomplete; this may affect our interpretations.
Despite these limitations, our study carries some strengths. We did not apply any publication year restriction to broaden our findings and sufficiently cover various patients from different geographical areas. We also exclude participants with major cardiac comorbidities and pregnancy, which may affect the incidence of thyrotoxic TR and/or MR, in order to obtain causality explanations regarding the pathophysiology.
Conclusion
The results of this systematic review show that with adequate and aggressive therapy for thyrotoxicosis, thyrotoxic TR and/or MR can be improved more significantly in terms of their severity. This is dependent on hyperthyroidism control, especially in the first 6 months of therapy. A similar finding also applies to HF and PH.
Detailed history-taking and physical examination are crucial to determining the progression of thyrotoxic TR and/or MR and the effects of this on cardiorespiratory dysfunction. Furthermore, the appropriate treatment for each patient needs to be carefully decided depending on their individual clinical presentations to obtain the maximum clinical improvement.
Several mechanisms are involved in thyrotoxic TR and/or MR, including the direct TH effect and the indirect effect of other factors associated with hyperthyroidism. Through this study, we suggest that future studies comparing thyrotoxic patients with and without TR and/or MR are warranted to clarify our findings and to establish the relationship between thyrotoxicosis and valvular regurgitation, mainly TR and MR.
Clinical Perspective
- 83.3% of thyrotoxic patients with tricuspid regurgitation (TR) improve clinically within 6 months of follow-up
- 69.23% of thyrotoxic patients with mitral regurgitation (MR) improve clinically in 3 months of follow-up
- 66.67% of heart failure patients and 76.19% of those with pulmonary hypertension improve clinically in 2–6 months of follow-up
- Aggressive therapies are needed to improve the severe thyrotoxic TR and MR.
- Direct and indirect thyroid hormone effects are involved in thyrotoxic TR and MR.