AF is a significant global public health concern, affecting an estimated 60 million individuals worldwide in 2019.1 In addition to the well-established risk of stroke, there has been increasing recognition of systemic comorbidities and complications associated with AF, including deterioration of renal function, which have garnered attention and have been addressed in clinical guidelines.2–4 AF is associated with an elevated risk of impaired renal function, which can occur through several mechanisms, including decreased renal blood flow due to activation of the renin–angiotensin–aldosterone system and the formation of microthrombi, leading to renal microinfarction.5–7 Furthermore, there is concern over the prolonged usage of oral anticoagulants in patients with AF, particularly vitamin K antagonists (VKAs), which accelerate the progression of renal disease.8
The effect of direct oral anticoagulants on kidney function has recently gained attention, particularly through the real-world data.9 The renal benefits of direct oral anticoagulants, particularly rivaroxaban and dabigatran, are acknowledged in AF management guidelines.2 However, owing to its high degree of renal excretion, dabigatran is less favoured for patients with renal impairment.4,10 In contrast, rivaroxaban is considered a preferred option for patients with renal impairment or underlying concerns about impaired renal function due, in part, to its reduced susceptibility to changes in kidney function.4,10 In addition, the potential benefits of rivaroxaban on kidney function further support its preferred use in this patient group.4,10 Nonetheless, assessment of the effects of rivaroxaban on renal outcomes relative to VKAs has predominantly relied on individual real-world evidence studies, with a paucity of comprehensive assessment. Therefore, a systematic review and meta-analysis are warranted to provide more certain evidence of the effects of rivaroxaban on renal outcomes in AF patients, particularly among subpopulations at elevated risk of unfavourable renal events.
The aim of this systematic review and meta-analysis was to provide a comprehensive comparative assessment of the effects of rivaroxaban and VKAs on renal outcomes in AF patients. This encompasses evaluation of acute kidney injury (AKI) and deterioration of renal function. The analysis includes subgroup analyses that focus on specific high-risk populations, such as the elderly (age ≥65 years), individuals with diabetes and those with pre-existing renal conditions, thus enabling a more nuanced evaluation of the data. Moreover, the analysis provides valuable insights into the effects of rivaroxaban and VKAs in the Asian population.
Methods
Protocol and Registration
This systematic review and meta-analysis adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.11 The protocol was collaboratively drafted by three authors (SHV, HPM, PPD) and was comprehensively reviewed and approved by all authors. The protocol has been registered and published on PROSPERO (ID: CRD42023462756).
Search strategy
The data search was conducted across three databases, namely PubMed, EMBASE and the Cochrane Library, from 1 January 2017 to 30 June 2023. The search used the following keywords and Medical Subject Headings (MeSH): ‘atrial fibrillation’, ‘rivaroxaban’, ‘acute kidney injury’, ‘≥30% decreases in estimated glomerular filtration rate’, ‘doubling of the serum creatinine level’, ‘progression to end-stage renal disease’, ‘requirement for haemodialysis’ and ‘need for a kidney transplant’. Our review was restricted to records published in the English language. Additional details of the search strategy are provided in Supplementary Table 1.
Inclusion and Exclusion Criteria
Real-world evidence studies that compared the effects of rivaroxaban and VKAs on desired renal outcomes in patients with AF were included in the analysis. Studies with a limited total patient sample size (<500 patients) and inadequate anticoagulation duration (<6 months) were excluded from the analysis. Two authors (HVS, PPD) conducted the data search in accordance with a predetermined protocol. They screened the records, identified eligible studies and extracted data. In case of disagreements, resolution was sought through consultation with all authors, with any remaining issues decided by a third author (HPM).
Outcomes
Four distinct kidney-related outcomes were assessed: AKI; a ≥30% decreasing in the estimated glomerular filtration rate (eGFR); doubling of the serum creatinine concentration; and worsening renal function, defined as a composite outcome encompassing kidney failure, progression to end-stage renal disease (eGFR ≤15 ml/min/1.73 m2) and the need for haemodialysis or a kidney transplant.
In addition to the pooled analysis, we conducted subgroup analyses on predefined outcomes. These subgroups comprised patients with diabetes, those with pre-existing kidney disease, the elderly (age ≥65 years) and Asian populations.
We used the ROBINS-I tool to assess the risk of bias. Review Manager (RevMan), a web-based platform for reviews, was used to facilitate the assessment. Two independent reviewers (HVS, PPD) evaluated bias risk, resolving disagreements through consultation with all authors, with any remaining issues decided by a third author (HPM).
Statistical Analysis
Data synthesis encompassed the use of HRs and corresponding 95% CIs as the primary effect size estimates. We used a random-effects model to account for potential heterogeneity among studies. To determine the relative importance of individual study effect size estimates, the inverse variance method was used. Heterogeneity was assessed using the Χ2 test (with p<0.05 indicating potential heterogeneity) and the I² statistic (which quantifies the percentage of total variation across studies due to heterogeneity). I² values of 25%, 50% and 75% are interpreted as indicating low, moderate and high heterogeneity, respectively. In addition, the amount of heterogeneity was quantified using the τ² statistic. Data analysis was conducted using the web-based RevMan, which is a universally recognised platform for conducting systematic reviews and meta-analyses.
Sensitive Analysis
Sensitivity analyses were performed to evaluate the robustness of the results and to assess the effects of methodological choices. In our Model 1 sensitivity analysis, we evaluated the effects of diabetes, pre-existing renal disease, Asian ethnicity and dosing strategy on the effects of rivaroxaban on renal outcomes to ascertain the robustness of our findings across these diverse patient subgroups. In Model 2, conference abstracts were excluded due to data limitations.
Results
Search Results and Study Characteristics
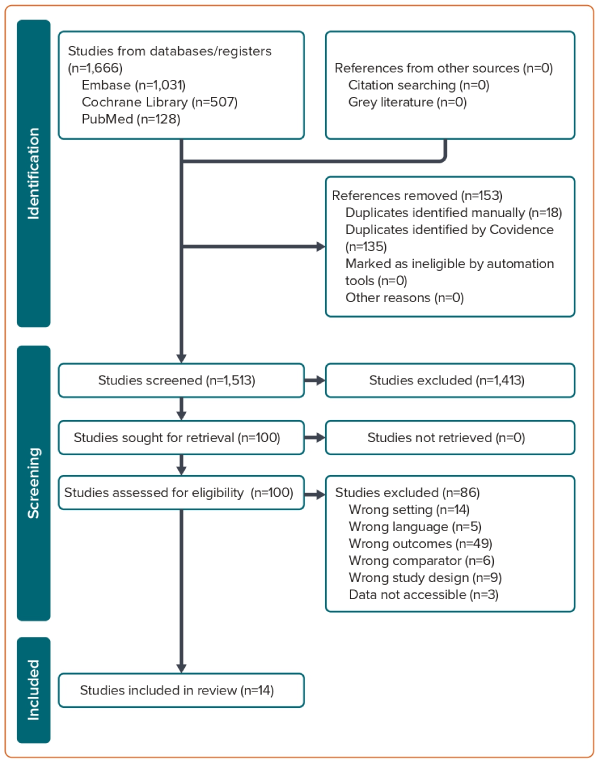
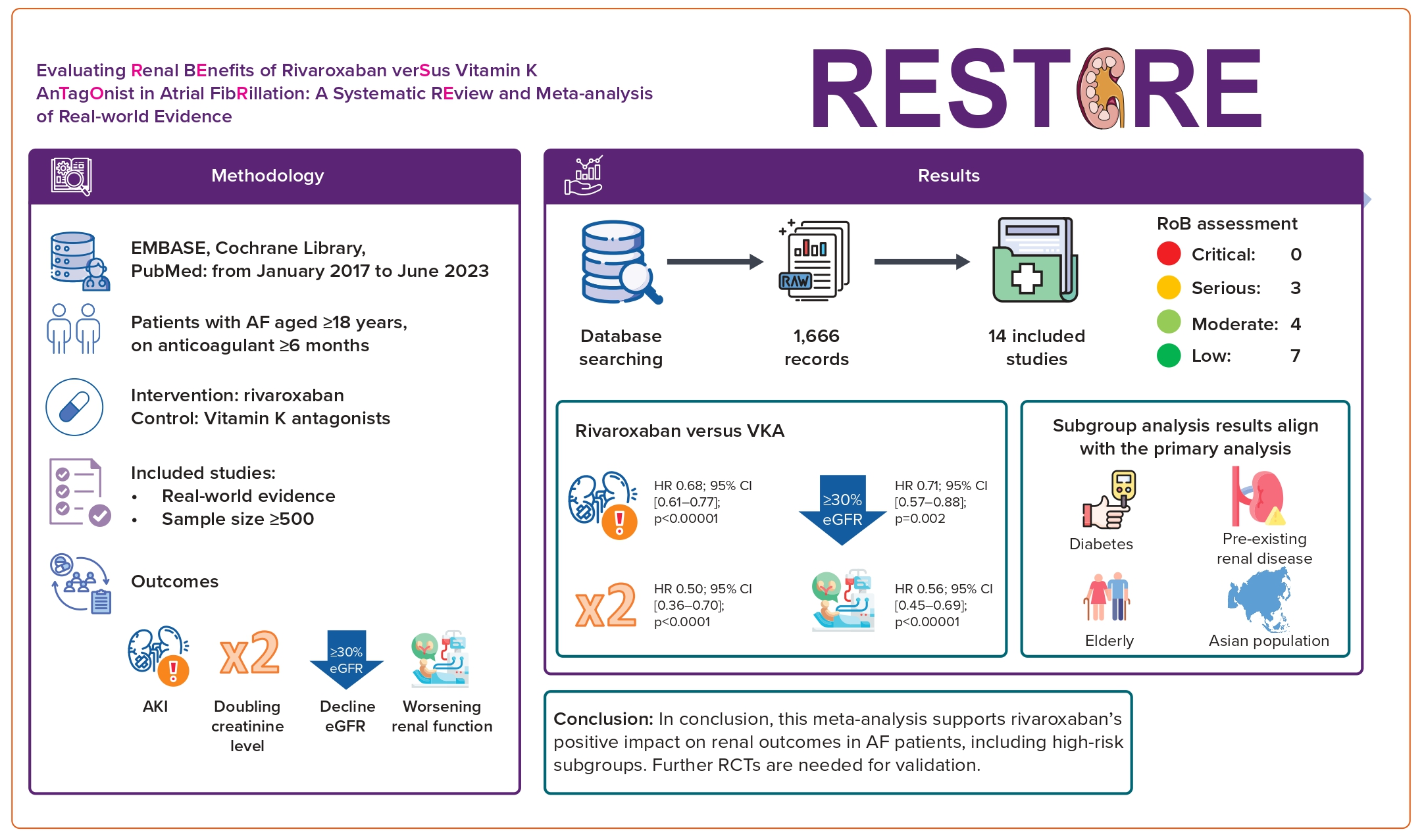
We initially identified 1,666 records from the PubMed, EMBASE and the Cochrane Library databases. Screening of titles and abstracts resulted in the exclusion of 1,564 records: 153 duplicates and 1,413 that were not relevant. Of the 102 records subjected to full text review, 86 were excluded for various reasons. Thus, 14 studies that met the inclusion criteria were selected for analysis. A flow chart of the selection process is shown in Figure 1.
A comprehensive analysis was conducted on 14 studies, involving a total cohort of 418,201 patients with AF.9,12–24 Of these 418,201 patients, 142,494 received rivaroxaban and 196,337 received VKAs. The study conducted by Klil-Drori et al. lacks information regarding the specific number of patients using rivaroxaban and VKAs.20 Thirteen of the 14 studies included in the analysis were observational and retrospective in nature, with the study by Kreutz et al. standing out as the sole prospective observational study.21 Importantly, four of the 14 studies were presented in abstract form at a conference.12,13,20,21
Most of the studies included in the analysis were well designed and used methods that matched clinical characteristics and potential confounders. The method used to address potential confounders across the studies varied. Specifically, 12 of 14 studies used propensity score weighting, with the most common being inverse probability of treatment weighting (IPTW).9,12–16,18,19,21–24 González et al. used Cox proportional hazards regression to adjust for confounding factors.17 Notably, the study conducted by Klil-Drori et al. did not provide information as to the methods used to adjust for confounding factors.20
Details of the included studies are provided in Supplementary Table 2.
Clinical Outcomes
Acute Kidney Injury
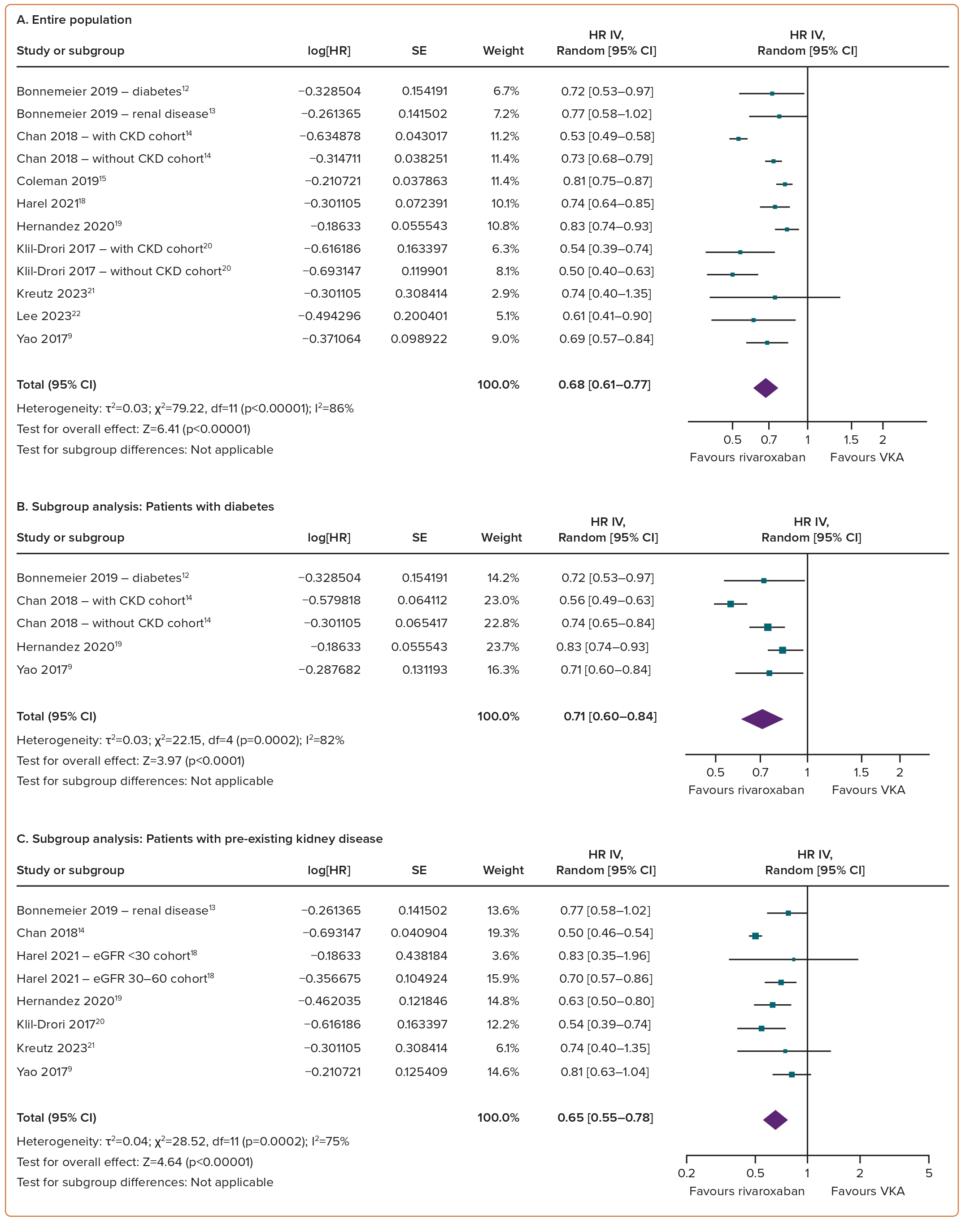
Compared with VKAs, the use of rivaroxaban in AF patients was linked to a significant reduction in the risk of AKI (HR 0.68; 95% CI [0.61–0.77]; p<0.00001; I²=86%; Figure 2A).
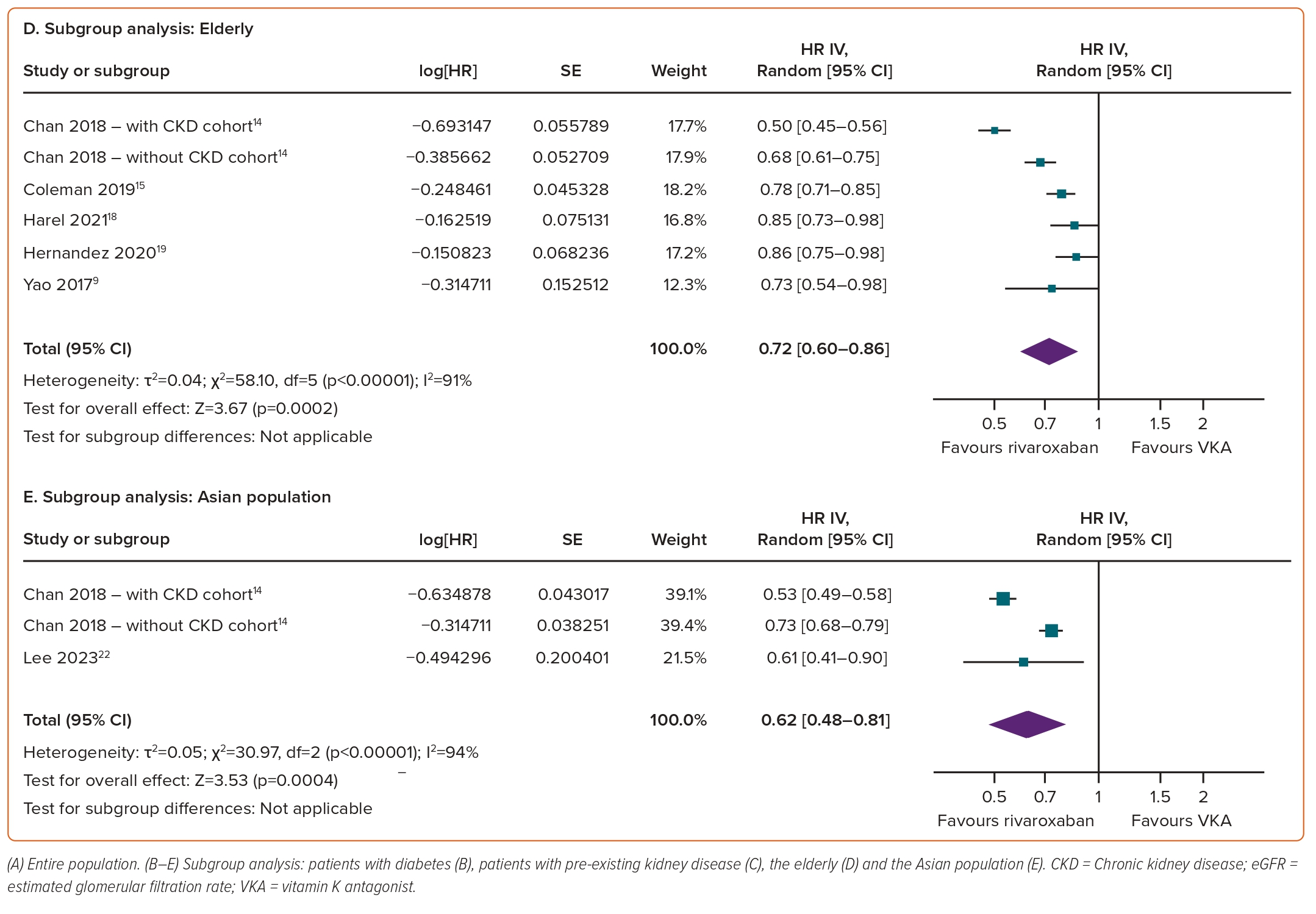
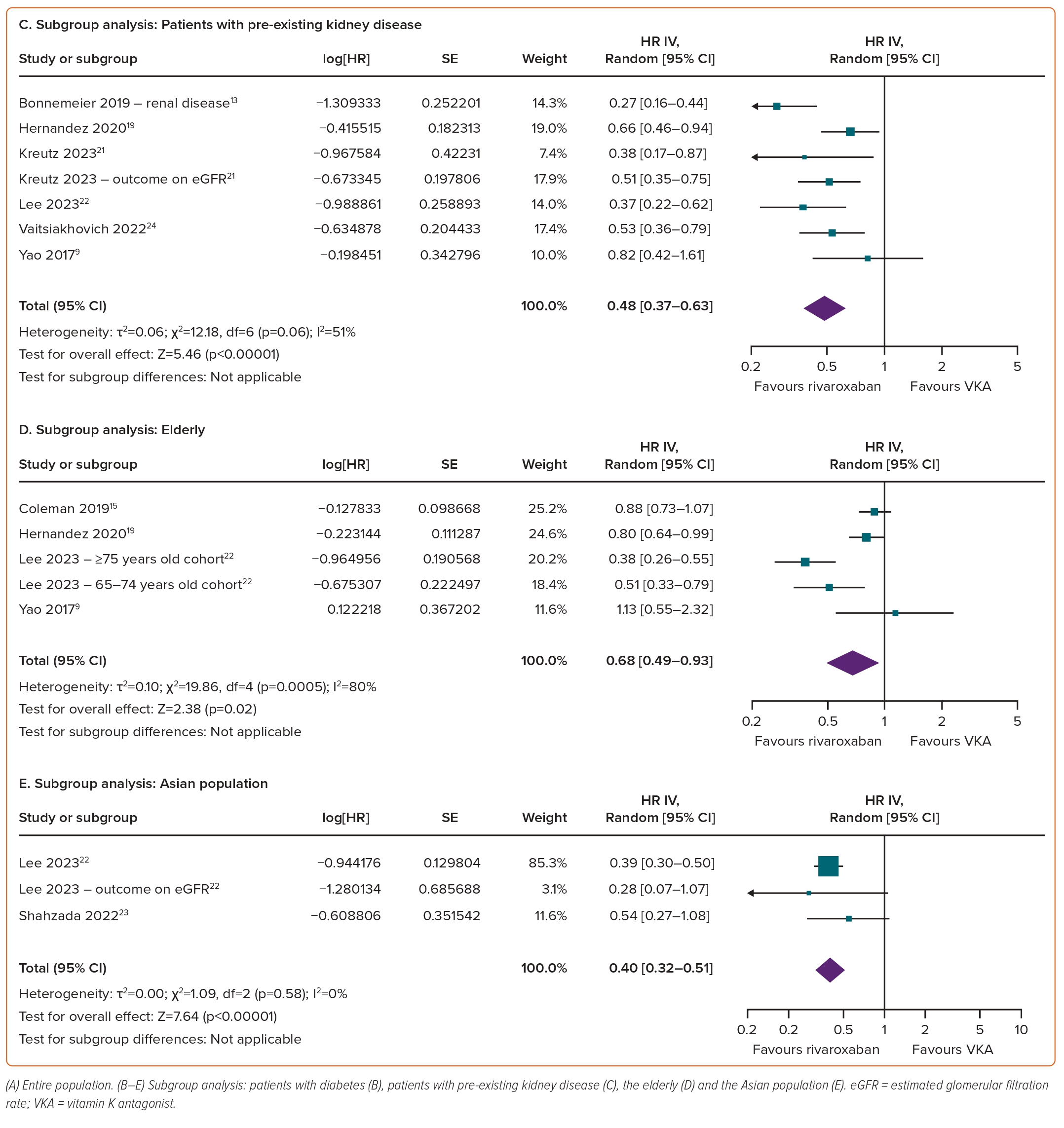
The results of prespecified subgroup analyses (diabetes, pre-existing kidney disease, the elderly and Asian populations) consistently highlighted the superiority of rivaroxaban over VKAs with regard to AKI outcomes across the various subgroups. Specifically, rivaroxaban was associated with a significantly reduced risk of AKI in individuals with diabetes (HR 0.71; 95% CI [0.60–0.84]; p<0.0001; I²=82%; Figure 2B), pre-existing kidney disease (HR 0.65; 95% CI [0.55–0.78]; p<0.00001; I²=75%; Figure 2C), the elderly (HR 0.72; 95% CI [0.60–0.86]; p=0.0002; I²=91%; Figure 2D) and the Asian population (HR 0.62; 95% CI [0.48–0.81]; p=0.0004; I²=94%; Figure 2E).
Decrease (≥30%) in Estimated Glomerular Filtration Rate
Compared with VKAs, the use of rivaroxaban in AF patients was linked to a significant reduction in the risk of a ≥30% decreased in eGFR (HR 0.71; 95% CI [0.57–0.88]; p=0.002; I²=95%; Figure 3A).
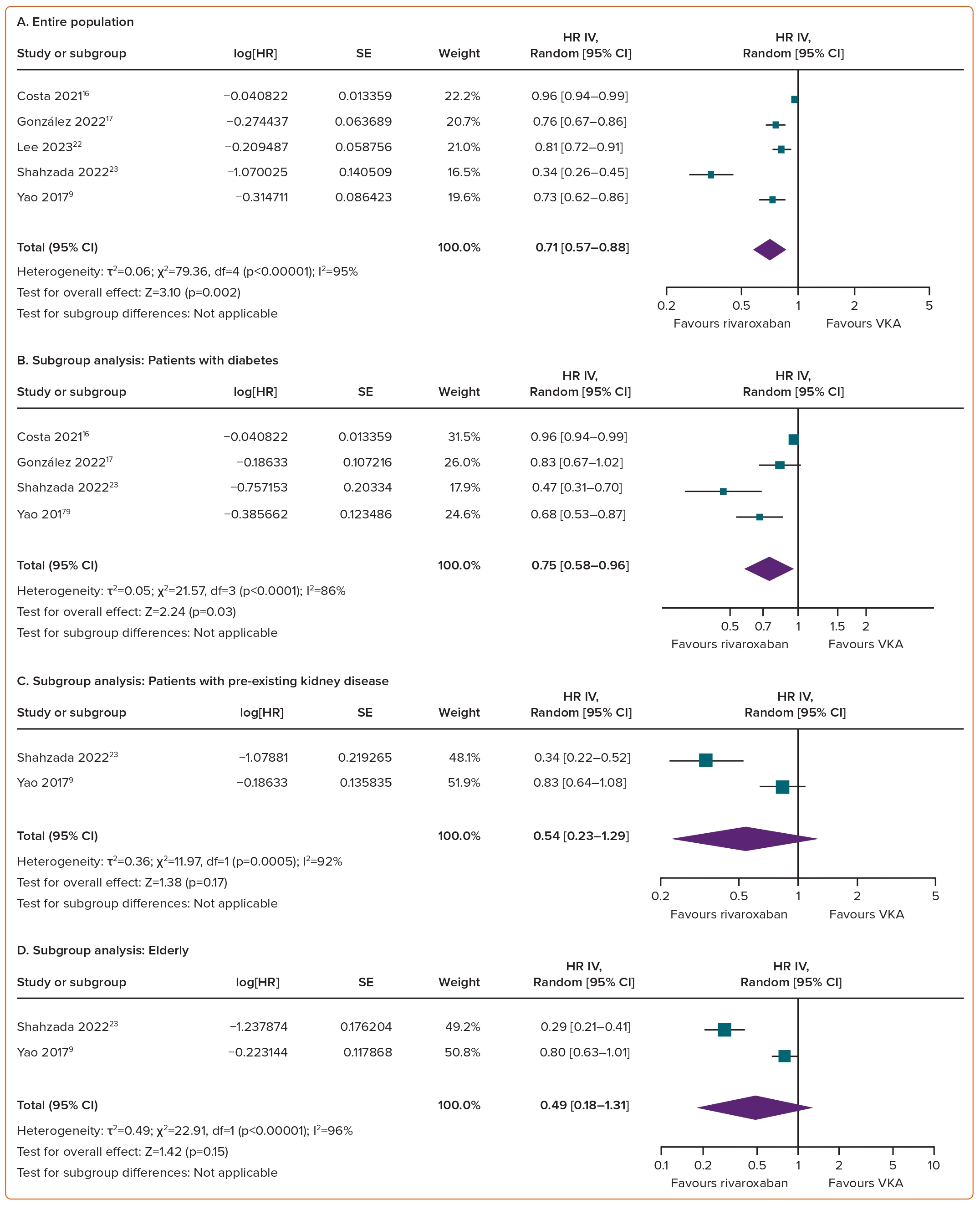
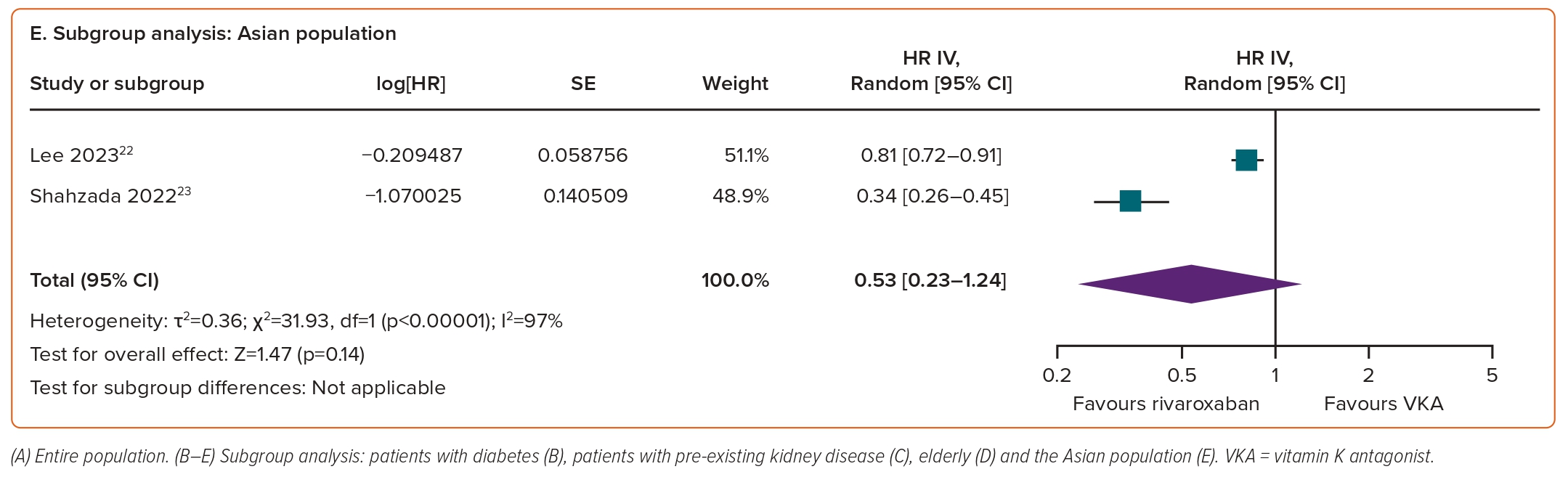
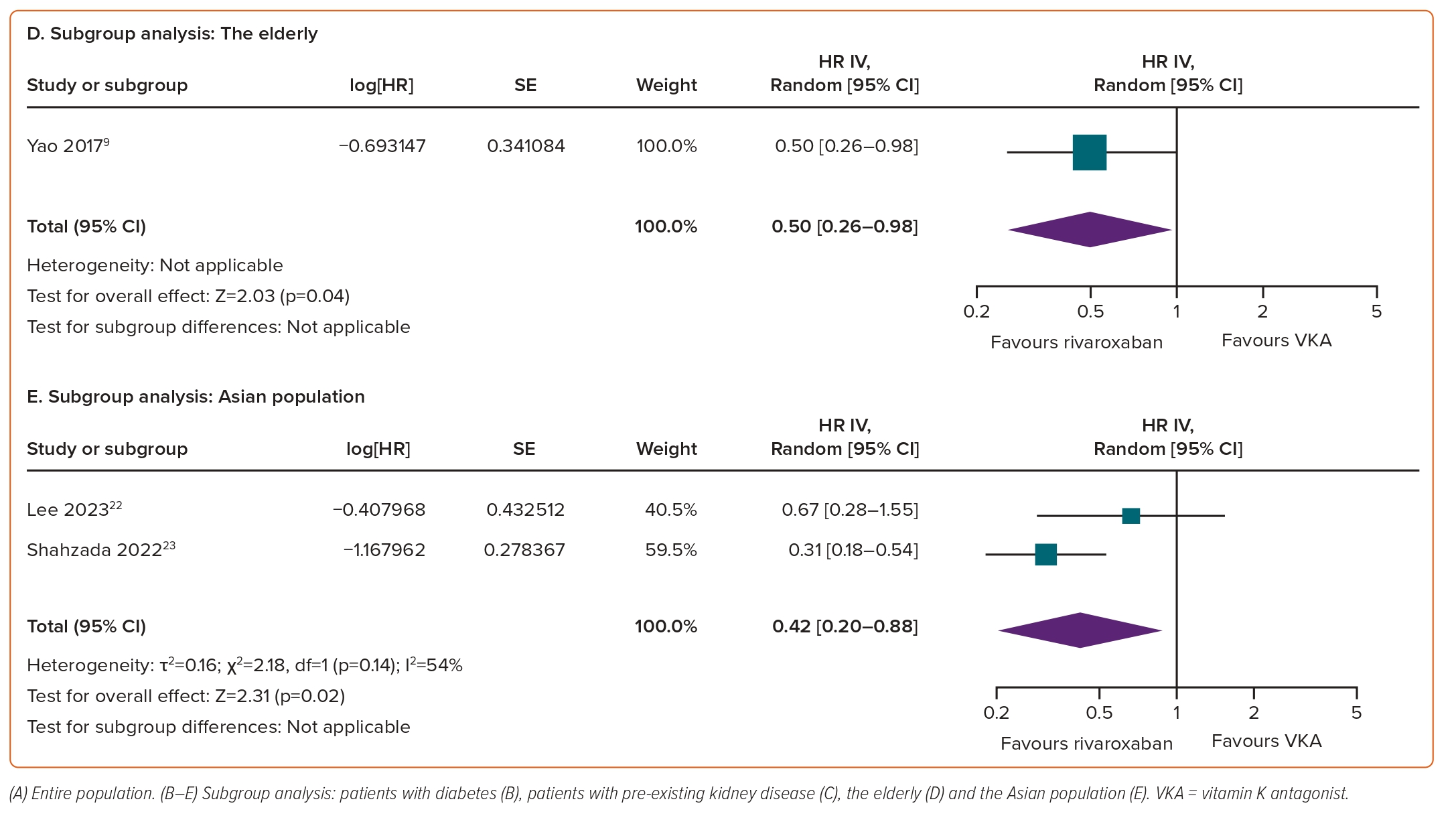
In subgroup analyses, rivaroxaban significantly reduced the risk of risk of a ≥30% decrease in eGFR in individuals with diabetes (HR 0.75; 95% CI [0.58–0.96]; p=0.03; I²=86%; Figure 3B). For the remaining three subgroups, there was a trend towards a positive effect of rivaroxaban on declining eGFR outcomes; however, the differences versus VKAs did not reach statistical significance, with HRs of 0.54 (95% CI [0.23–1.29]; p=0.17; I²=92%; Figure 3C) for pre-existing kidney disease, 0.49 (95% CI [0.18–1.31]; p=0.15; I²=96%; Figure 3D) for the elderly and 0.53 (95% CI [0.23–1.24]; p=0.14; I²=97%; Figure 3E) for the Asian population.
Doubling of the Serum Creatinine Concentration
Compared with VKAs, the use of rivaroxaban in AF patients was associated with a reduced risk of doubling of the serum creatinine concentration (HR 0.50; 95% CI [0.36–0.70]; p<0.0001; I²=50%; Figure 4A).
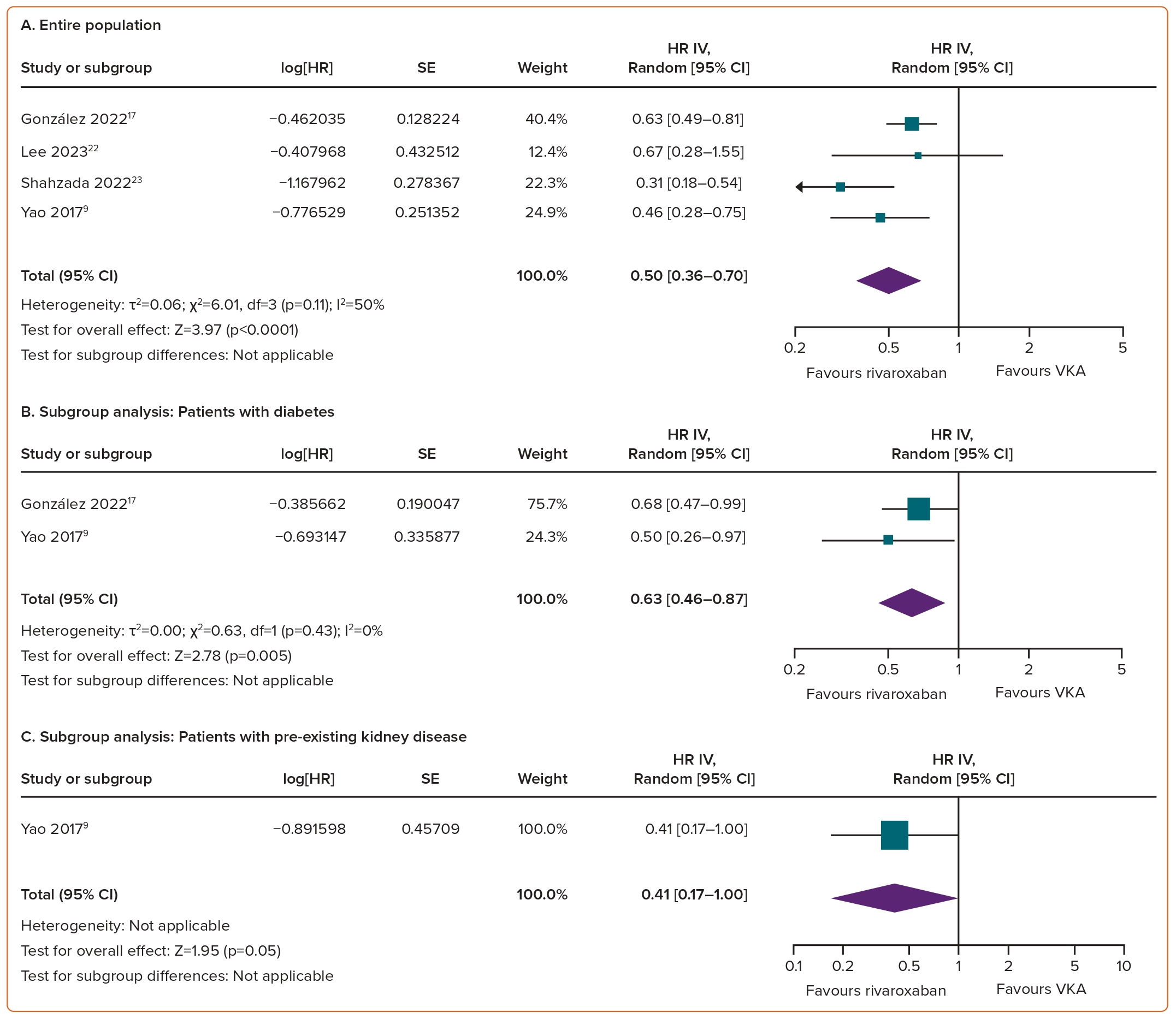
Worsening Renal Function
Compared with VKAs, the use of rivaroxaban in AF patients was associated with a reduced risk of worsening renal function (HR 0.56; 95% CI [0.45–0.69]; p<0.00001; I²=89%; Figure 5A).
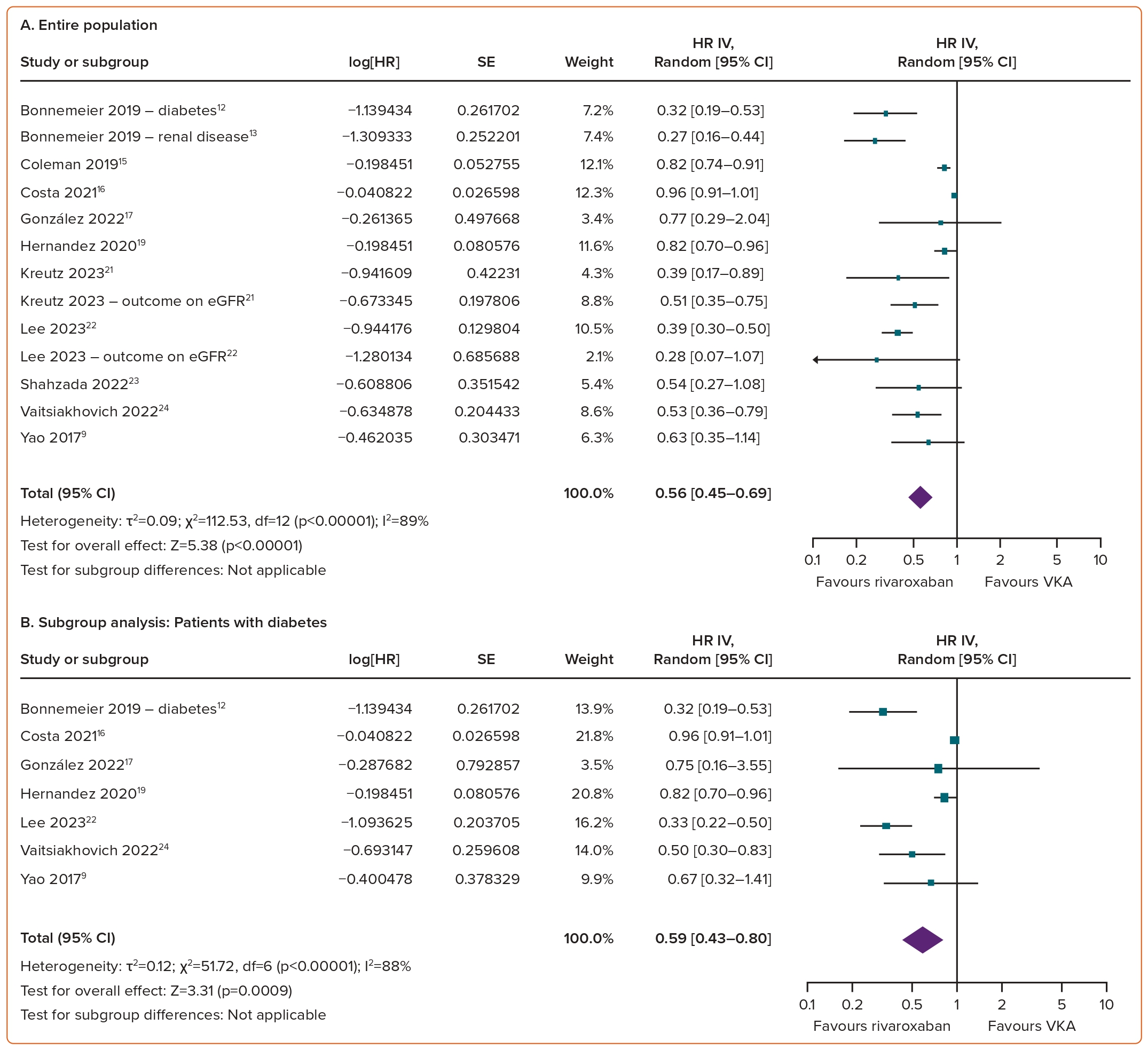
Consistent results across all subgroups underscore the superiority of rivaroxaban over VKAs in terms of worsening renal function. Rivaroxaban significantly reduced the risk of renal function worsening in individuals with diabetes (HR 0.59; 95% CI [0.43–0.80]; p=0.0009; I²=88%; Figure 5B), pre-existing kidney disease (HR 0.48; 95% CI [0.37–0.63]; p<0.00001; I²=51%; Figure 5C), the elderly (HR 0.68; 95% CI [0.49–0.93]; p=0.02; I²=80%; Figure 5D) and the Asian population (HR 0.40; 95% CI [0.32–0.51]; p<0.00001; I²=0%; Figure 5E).
Risk of Bias
Most studies analysed were well designed, using methods to equitably balance clinical characteristics and adjust for confounding factors. Seven studies had a low risk of bias,9,12,13,16,17,21,23 whereas four studies had a moderate risk.14,15,19,24 Of the three studies with a serious risk of bias, those conducted by Harel et al. and Lee et al. were identified as having a serious risk of bias in the missing data domain, with the remaining domains showing low risk of bias.18,22 The study by Klil-Drori et al. lacked clarity in several domains.20
Details of the risk of bias assessment are provided in Supplementary Table 3. The funnel plot for assessing publication bias is shown in Supplementary Figure 1.
Sensitive Analyses
We conducted sensitivity analyses to evaluate the robustness of the results and to examine the effects of various methodological choices or assumptions on the overall findings. In Model 1, we assessed the effects of diabetes, renal disease, dosing strategy and race on the effects of rivaroxaban on renal outcomes compared with VKA. In Model 2, we excluded the conference abstracts due to limited data accessibility, particularly the two studies by Bonnemeier et al. because of potential partial overlap in the study populations.12,13,20,21
The sensitivity analysis Model 1 revealed that the relative effects of rivaroxaban compared with VKA on renal outcomes were consistent across most examined subgroups. However, a notable exception was observed within the Asian population subgroup, where the beneficial effect of rivaroxaban on worsening renal function was more pronounced than in the non-Asian population, with HRs of 0.40 (95% CI [0.32–0.51]; p<0.00001) for the Asian subgroup and 0.61 (95% CI [0.50–0.75]; p<0.00001) for non-Asian subgroup (pinteraction=0.0007). In Model 2, consistent findings emerged confirming the favourable effects of rivaroxaban relative to VKAs concerning kidney outcomes. Rivaroxaban demonstrated superiority in preventing AKI (HR 0.71; 95% CI [0.61–0.81]; p<0.00001; I²=91%), a ≥30% decrease in eGFR (HR 0.71; 95% CI [0.57–0.88]; p=0.002; I²=95%), doubling of the serum creatinine concentration (HR 0.50; 95% CI [0.36–0.70]; p<0.0001; I²=50%) and deterioration of renal function (HR 0.89; 95% CI [0.85–0.93]; p<0.00001; I²=88%).
The forest plot of sensitive analyses is provided in Supplementary Figure 2 and Supplementary Table 4
Discussion
To the best of our knowledge, this is the first systematic review and meta-analysis comparing rivaroxaban and VKAs on four major renal outcomes, namely AKI, a ≥30% decrease in eGFR, doubling of the serum creatinine concentration and worsening renal function. Anticoagulant-related nephropathy, initially identified as a manifestation of AKI, occurs in patients exposed to excessive anticoagulation with VKAs (international normalized ratio of prothrombin time [INR] >3).25 AKI may manifest in about one-fifth of patients with supratherapeutic levels of VKAs (INR >3) within a week.26 Hence, the assessment of incident AKI serves to facilitate a comparative analysis of the effects of rivaroxaban and VKAs on early-stage renal injury. Endpoints of a 30–40% decline in eGFR and doubling of the serum creatinine concentration are endorsed by the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines for evaluating progression of renal disease in the context of clinical trials.27 Meanwhile, the endpoint of worsening renal function aims to evaluate the long-term effects of anticoagulation on renal outcomes. The constituent outcomes incorporated into this composite measure align with recommendations from KDIGO, the National Kidney Foundation and the US Food and Drug Administration for conducting clinical trials on nephropathy.27,28 We believe that using a comprehensive set of clinical endpoints facilitated a thorough comparison of the effects of rivaroxaban and VKAs on renal outcomes in AF patients.
The results of the meta-analysis demonstrated the superiority of rivaroxaban over VKAs across all criteria evaluated. Moreover, we identified specific populations at heightened risk of adverse renal events with anticoagulant use, including the elderly, diabetic patients and those with pre-existing renal disease.26 Subgroup analyses were conducted on these subpopulations to provide a more nuanced assessment. In addition, the supratherapeutic levels with VKAs emerged as a well-established risk factor for anticoagulation-related kidney injury.29 Notably, our analysis also revealed that Asian patients exhibit poor INR control when undergoing anticoagulation, prompting a subgroup analysis for this population.30 The outcomes of the subgroup analyses consistently support the findings of the primary analysis, providing additional evidence for the renoprotective effects of rivaroxaban compared with VKAs.
Several potential mechanisms of action have been proposed to explain the differences in renal outcomes between rivaroxaban and VKAs. First, there are many pathways linking vascular calcification and arterial stiffness, which are processes correlated with low vitamin K levels, to chronic kidney disease.31 This may explain the deterioration in renal function observed in patients with AF when treated with VKAs.8,32 Unlike VKAs, rivaroxaban has no effect on the metabolism of vitamin K and may not adversely affect renal function through this mechanism. Conversely, switching from warfarin to rivaroxaban resulted in a significant decrease in pulse wave velocity, indicating an improvement in vascular stiffness associated with the use of rivaroxaban.33 Second, in individuals with AF, ischaemic damage and oxidative stress play notable roles in the pathogenesis of renal disease.7,34 Rivaroxaban exerts renoprotective effects by mitigating oxidative stress, which is a key contributor to kidney damage during ischaemia–reperfusion events. Rivaroxaban significantly reduces levels of malondialdehyde, a marker of oxidative stress, suggests that it helps preserve kidney function by limiting the harmful effects of free radicals and inflammation.35 By inhibiting Factor Xa, rivaroxaban not only prevents clot formation, but also appears to have anti-inflammatory actions that further protect renal tissues against damage associated with reperfusion after ischaemic episodes.35 In addition, rivaroxaban inhibits oxidative stress and preserves mitochondrial function in the kidneys. Rivaroxaban has been shown to decrease the production of reactive oxygen species, which are known to contribute to cellular damage and kidney dysfunction.36 By maintaining the integrity of mitochondrial function, rivaroxaban helps avert cell death pathways that can lead to renal injury.36 The protective effects of rivaroxaban are dose dependent, with higher doses proving more efficacious in reducing oxidative stress and maintaining mitochondrial health, both of which are critical for kidney function.36 Third, in the setting of hypertensive renal damage in hypertensive mice overexpressing renin, rivaroxaban significantly reduced albuminuria and attenuated histological changes, such as glomerular hypertrophy and mesangial matrix expansion. Rivaroxaban also inhibited glomerular basement membrane thickening and glomerular hypertrophy, demonstrating anti-inflammatory effects.37 Rivaroxaban exerts protective effects against angiotensin II-induced renal damage and protects against podocyte injury.37 These protective effects are due, in part, to the inhibition of the protease activated receptor-2 signalling-mediated inflammatory response.37 In a randomised trial involving patients with chronic kidney disease, rivaroxaban (compared with warfarin) reduced urinary albumin excretion and attenuated tubular injury in individuals with AF and micro-/macroalbuminuria chronic kidney disease.38
The primary limitation of our meta-analysis is the high heterogeneity among studies. The high heterogeneity in outcomes, specifically for AKI, a ≥30% decrease in eGFR and worsening renal function, warrants a multifaceted exploration of potential underlying factors. First, the variability in outcome definitions across the studies included is a salient contributor to heterogeneity. For example, Coleman et al. used International Classification of Diseases, Ninth and Tenth Revision diagnosis codes to define AKI, whereas Harel et al. used a clinical definition encompassing a ≥50% increase in serum creatinine concentration from baseline, an increase of ≥0.3 mg/dl or initiation of acute dialysis.15,18 The use of a composite definition for worsening renal function in our analysis, which amalgamated different outcomes of advanced stages of kidney injury, may have amplified the heterogeneity. Moreover, the heterogeneity was potentially exacerbated by the diversity in baseline renal function parameters, such as serum creatinine and eGFR, across the studies. Second, there is pronounced heterogeneity within the study populations themselves. The studies included encompass cohorts with distinct clinical characteristics, including (but not limited to) diabetes, pre-existing kidney disease and advanced age. These factors inherently influence renal outcomes and, consequently, may have contributed to the heterogeneity observed. Third, heterogeneity may be influenced by variations in anticoagulation management, including differences in dosing and treatment duration, as well as disparities in study design. We conducted a sensitivity analysis to determine the consistency of the renal outcomes with rivaroxaban. Our findings indicate that the effects of rivaroxaban on renal outcomes remain consistent regardless of patients’ diabetes status, the presence of pre-existing renal disease or the dosing strategy used. Notably, the benefit of rivaroxaban was more pronounced on worsening renal outcomes in the Asian subgroup, which may be attributed to the comparatively poorer INR control commonly reported in Asian populations. INR >3 have been associated with an increased risk of renal injuries in patients with AF, potentially explaining the enhanced efficacy of rivaroxaban in this population.26,39 The observed high heterogeneity underscores the need for careful consideration when interpreting the pooled outcomes. Robust randomised controlled trials are needed to confirm these results and provide a clearer understanding of the effects under more controlled conditions. Given the high heterogeneity, we used the random-effects model in our meta-analysis, acknowledging and accommodating the diverse methodologies among the studies included. In an effort to curtail the effect of publication bias, we conducted a comprehensive search across multiple databases and deliberately adopted inclusive selection criteria. In addition, we incorporated conference abstracts into our analysis to ensure the inclusion of pertinent but potentially unpublished studies. We excluded three studies due to data inaccessibility.40–42 However, their inclusion is unlikely to significantly alter the overall results. Conversely, our analysis incorporated four conference abstracts with limited data access. The results of a sensitivity analysis that excluded these four studies closely aligned with those of the primary analysis, reinforcing the robustness of our findings.
In the context of clinical implications, the findings of this systematic review and meta-analysis support the use of rivaroxaban in AF patients at an increased risk of deteriorating renal function. Specifically, these results endorse the use of rivaroxaban in individuals with diabetes, pre-existing kidney disease, the elderly with a heightened risk of renal injury and the Asian population.
Conclusion
In conclusion, this meta-analysis underscores rivaroxaban’s favourable effects on renal outcomes in AF patients. The findings from subgroup analyses of patients with diabetes, pre-existing kidney disease, the elderly and from the Asian population are consistent with the results of the primary analysis. Despite study limitations, these findings support rivaroxaban as the preferred anticoagulant to mitigate renal complications. The results offer clinicians valuable insights for tailored anticoagulation strategies, optimising renal outcomes. Further randomised control trials are warranted to validate the positive effects of rivaroxaban on renal outcomes.